Mobile App Monitoring in Gestational Diabetes Mellitus

Introduction
Gestational diabetes mellitus (GDM) commonly occurs during pregnancy and usually disappears once labour is completed, but some women will continue to have high blood glucose levels after delivery1. During 2017 in Australia alone, there were more than 45,000 women diagnosed with GDM. Around 10% of pregnant women are estimated to develop GDM during their pregnancy1.
Alongside the generally increasing prevalence of GDM in certain populations1 , changes in diagnostic and screening criteria has resulted in a greater number of women being identified and managed by the healthcare system for this condition.2 The increasing number of women developing GDM is putting great pressure on health services, with many hospitals and health care providers struggling to provide timely support and services to these women3 . New technologies such as mobile apps, when integrated into management, may be one way to increase the efficacy and availability of healthcare services for these women. These tools have also shown potential for improving glycaemic management in adults with type 1 and type 2 diabetes.4
Mobile phones have portability, reliable internet connectivity, and increasing capacity to run complex apps, which makes them ideal tools in health services to collect personal information, provide personalised intervention, and potentially save time and cost when compared with standard health care models. The management of GDM includes optimisation of lifestyle, self-monitoring of blood glucose, intensive education and introduction of medication when warranted. Utilisation of mobile phone apps as part of the self-management of GDM could benefit women with the condition and also the health professionals who treat them.
The aim of this review is to investigate the perception of women with GDM and health professionals of mobile apps available for managing GDM.
Methods
Scoping reviews are useful for synthesizing research evidence and are often used to map key concepts that underpin research in existing literature in a given field in terms of their nature, features, and volume – particularly in novel fields of enquiry5 . In this scoping review, we used the Arksey and O’Malley framework5 , utilising the first five of their six stages:
Stage 1: Identifying the research questions
The research questions addressed in this review were as follows:
1) What are the mobile apps available for GDM?
2) What are the reported perceptions of mobile apps for GDM among women with GDM and healthcare professionals?
Stage 2: Identifying relevant studies
Although a scoping review is designed to cover a broad spectrum of literature, inclusion and exclusion criteria guided the search and helped to filter the literature. The scoping review included published peer-reviewed papers that were retrieved from the following electronic databases: CINAHL, Cochrane library, Medline, PsycINFO and PubMed. Reference chaining (a review of links found through the electronic search) was undertaken to ensure that all possible relevant articles could be included in the scoping review.
Based on the initial exploratory search, the authors agreed on the following eligibility criteria: include all peer-reviewed literature that explored the keywords identified in Table 1. To ensure a comprehensive search of the literature was undertaken, no date of publication criterion was applied. An academic librarian was consulted for advice on the most appropriate Medical Subject heading terms (MeSH) for the search and how to modify MeSH terms for the different databases used. Based on this extensive exploratory scoping phase, the search strings for each database were finalised.
Data such as conference abstracts, commentaries, books and book reviews, editorial articles and non-peer-reviewed grey literature, including health service policy and procedure documents, was accessed and read, but excluded. This enabled the literature used to inform these excluded sources to be accessed for inclusion (reference chaining). Papers not written in English were also excluded.
Stage 3: Study selection
The third stage of Arksey and O’Malley’s framework enables identification of studies to be included in the scoping review5 (See Figure 1). We consolidated the results of the searches from different databases and excluded duplicates. Two authors (MD, HW) then independently screened the titles and abstracts of the articles against the eligibility criteria. Disagreements about study eligibility were resolved with a third author (VN) by consensus discussion. Studies quality was assessed using Mixed Method Assessment Tool (MMAT)6 criteria based on the methodology used concomitantly with data extraction to evaluate the quality of the studies using various methodologies and subsequently establish their comparative validity and reliability. Quality scores ranging from 0% representing no criteria met, through to 100% representing all criteria met6 . Two researchers (MD, HW) independently appraised each article and then compared and discussed their final scores. As per scoping review methodology, quality scores were not used to exclude studies. The second part of the review produced six papers published between 2015-2018. One paper was published in the UK, three papers were published in Norway, and one each from New Zealand and Israel respectively. The included studies were split into either qualitative studies or randomised control trials. Study participants ranged from 5 to 120 women.
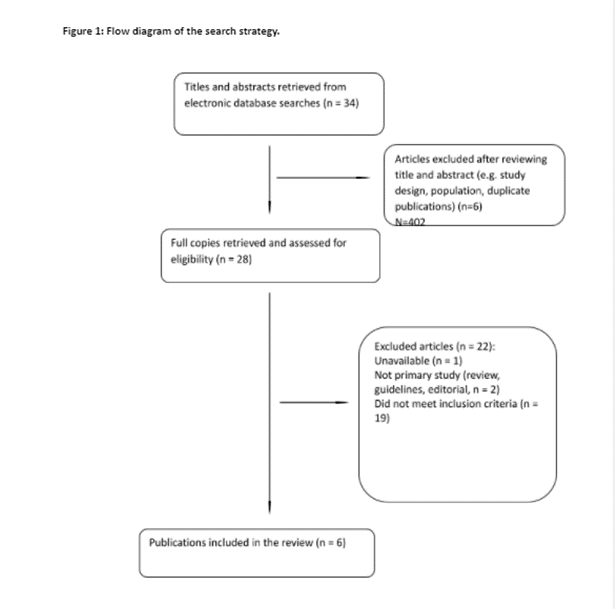
Stage 4: Charting the data
Based on the preliminary scoping phase, a data extraction framework was developed. The final concept map included nine categories that were used to extract data to address the study aim (see Table 2). The framework was pilot tested by two authors (MD, HW) on a sample of the included studies (i.e. 10% of the complete list of retrieved studies) in order to ensure that the coding framework was consistently applied.
Authors independently extracted the data from each included study to ensure inter-rater reliability. A sample (20%) of the included articles were independently reviewed and compared. Discrepancies in extracted data were discussed between the research team until consensus was reached.
Stage 5: Collating, summarising and reporting the results
Analysis and conceptual synthesis of the data collected using the data extraction framework provided information on the body of research undertaken around the aims.
Results
Two separate searches were conducted in order to achieve the aims of the review. Firstly, two app stores, (Apple and Google Play stores) were searched for apps relevant to women with GDM in order to establish what options were currently available for these women. A total of 19 mobile apps were found – 11 in the Apple Store, seven in the Google Play store and one app in both stores (see Table 2). While most apps were free, the paid apps ranged from $3-8 AUD. One app was a platform for women with GDM to share their experiences. Nine of the apps were developed to track blood glucose levels during pregnancy (with or without sharing data with the health professional), six apps were focused on providing information around GDM, two apps offered dietary tips for women with GDM and one app focused on managing stress during pregnancy with GDM. The number of downloads and reviews were recorded, where available, to give an indication of the relative popularity of these different options. Of the apps that had reviews available, they ranged from 1 to 5 (out of 5) stars with the main issues raised including limited information and difficulties with access. Positive written reviews included an improved ability to evaluate stress after using the Gestational Diabetes Stress Scale app.
All six of the studies described the utilisation of a mobile app to monitor blood glucose levels, linked between a smartphone and a blood glucose meter via Bluetooth. One of the studies additionally looked at the apps that supported lifestyle modifications in women with GDM7. The mobile app, Pregnant+ (which records blood glucose readings via its Bluetooth connection and allows for this data to be shared with health professionals) was investigated in a series of trials from Norway8, 9 . Two studies investigated the experiences of both women with GDM and health professionals in utilising these apps8, 7 . Overall, three studies focused on feedback from health professionals and five studies obtained feedback from women with GDM. These findings are summarised in Table 3.
Hirst, Mackillop et al10 conducted a cohort study of 52 women with GDM using a Bluetooth connected blood glucose monitor which automatically recorded readings via a smartphone app. While the perceptions of healthcare professionals were not recorded in this study, it found that women with GDM reported overall satisfaction of with the useability of application. The majority of women provided positive feedback that the application was convenient and reliable.
Garnweidner-Homle, et al (2015 and 2018)8, 9 both focused on a mobile app permitting Bluetooth blood glucose monitoring and providing information regarding the management of GDM using three distinct focus groups, including two groups, health professionals (n=5 and n=9) and 21 women with GDM. The health professionals (midwives and diabetes educators), suggested that the app needed to be more GDM and client specific and that optimal blood glucose levels can be highly variable between individuals, making generic information less useful. However, they concluded that it was an appropriate tool for the care of women diagnosed with GDM given that it could provide healthcare professional approved information for women to access at home. Women with GDM reported that they would use the app in the future, particularly due to the ease in which it allowed them to record their blood glucose levels. One participant stated “I think it is very good. You get inspired to do things right. It’s pretty easy, yes, because you have your phone with you all the time. I always forget about the registration booklet and have to find a pen.”8
In their randomised controlled trial of 120 women with newly diagnosed GDM, Miremberg et al (2018)11 found that a smartphone app increased adherence to regular blood glucose monitoring, improved glycaemic management and reduced the need for insulin treatment. The majority of participants reported high levels of satisfaction with the use of the app and their overall prenatal care. Healthcare professional experience of using the application was not reported.
The review conducted by Pais et al (2017)7 resulted in the formation of a website where pooled, metabolic data from a number of monitoring apps utilised by participants could be accessed by both health professionals, and women with GDM. This included weight, diet and blood glucose readings. Both groups reported that such a system was useful in the management of GDM. Overall, health professionals (including two dieticians, a midwife, an obstetrician and a physician) found it useful to have metabolic data brought together with blood glucose readings while the five women with GDM in the study were enthusiastic about sharing wellness data with their respective health professionals. One health professional summarised these common themes by stating “Yes, it is including pretty much everything that you want to know about: blood glucose, food diary, exercise.” 7 They qualified this however, by suggesting that appropriate and accurate apps would need to be included particularly in areas of carbohydrate counting.
Finally, Skar et al (2018)12 found more mixed opinions regarding the utility of a mobile app for blood glucose monitoring. A subset of women in the trial (4 of 17) reported that the app promoted feelings of obsession and frustration, and that a limitation of the system was the inability of health professionals to assist with technical issues. Two of the women in the study additionally reported that in some instances, health professionals were unwilling to engage with usage of the app, rendering it obsolete in their management. The authors concluded that closer collaboration between healthcare professionals and the women using these apps would be necessary to see the most value gained from them12 . The overall findings however, suggested that the app was useful in promoting and maintaining behavioural change.
Table 1: Key words variation used in search strategy
| Patient | Health professional | Perception | Mobile app | Gestational diabetes |
| Patient* | Health professional* Clinician* Midwi* Nurs* Doctor* Physician* Dietitian* | Patients’ perception survey*; patients’ satisfaction survey*; patients’ feedback, feedback, patients’ opinion*, patients’ comment*, patients’ preference*, patients’ experience*, patients’ expectation*, questionnaire*, evaluation*, (consumers* was also used to replace search terms with Patients*) | Mobile app* | Gestational diabetes* |
Table 2: iTunes app and Google Play store search.
| No. | App name (Focus) | Downloads | Cost (AUD) | Review/Score | App Store |
| 1 | Diabetes Forum (Discussion/Forum) | N/A | Free | 4.0 (5 ratings) | Apple |
| 2 | FETAL LIFE (BS tracking, data sharing) | N/A | Free | N/A | Apple |
| 3 | Gestational Diabetes (information) | 100+ | Free | N/A | Google Play |
| 4 | gestational diabetes/information (education) | 1k+ | Free | N/A | Google Play |
| 5 | Gestational Diabetes (Diet/information/education) | 10k+ | Free | 1.8 (20 ratings) “…It only linked to websites with very little information…” | Google Play |
| 6 | Gestational Diabetes Food (advice, tips) | N/A | $4.49 | N/A | Apple |
| 7 | Gestational Diabetes Health Tips and Care(information/education) | 100+ | Free | N/A | Google Play |
| 8 | Gestational Diabetes Meal (dietary advice) | 100+ | Free | N/A | Google Play |
| 9 | Gestational Diabetes Stress Scale (stress/monitoring) | 1k+ | Free | 4.5 (54 ratings) “Very useful for GDM patients, to evaluate their stress in GDM.” | Google Play |
| 10 | Glucolyf (BS tracking, data sharing) | N/A | Free | N/A | Apple |
| 11 | GlucoKeeper (BS tracking) | N/A | Free | N/A | Apple |
| 12 | Glucose – Blood Glucose Tracker (BS tracking) | N/A | Free | 4.6 (281 ratings) | Apple |
| 13 | gluQUO: Control your diabetes (management, information) | N/A | Free | N/A | Apple |
| 14 | Glycemic Diary: Manage Diabetes (BS tracking) | N/A | $2.99 | N/A | Apple |
| 15 | Habits: Gestational Diabetes (BS tracking, data, sharing with health professionals) | N/A | Free | 1.0 (three ratings) “Can’t use it unless your midwife gives you an access code.” | Apple/Google Play store |
| 16 | moTHer (BS, health metric tracking) | N/A | Free | 2.3 (three ratings) | Apple |
| 17 | MyFetalLife (BS tracking, data sharing) | N/A | Free | N/A | Apple |
| 18 | Pregnant with diabetes (Information) | N/A | Free | 5.0 (one rating) from iTunes App 3.6 (43 ratings) “This app is purely definitions, no actual support for pregnant diabetics” | Apple |
| 19 | What To Do With Gestational Diabetes(information/education) | N/A | $7.99 | N/A | Google Play |
Table 3: Concept Mapping, Studies Included for Final Analysis.
| Reference | Country | Study Type | Participant no | Period of Utilisation | App and technology characteristics | GDM management focus |
| 10 | UK and Australia | Cohort study | 52 | 8-10 weeks | A Bluetooth connected blood glucose monitor automatically records readings via the smartphone app | Blood glucose monitoring |
| 8 | Norway | Focus Group | 21 women with GDM 5 Health professionals | Two, month-long testing periods | Permits Bluetooth blood glucose monitoring and provides information regarding the management of gestational diabetes | Blood glucose monitoring. Lifestyle modifications |
| 9 | Norway | Focus Group | 9 | Interviews lasted between 16-35 minutes | Permits Bluetooth blood glucose monitoring and provides information regarding the management of gestational diabetes | Blood glucose monitoring. Lifestyle modifications |
| 11 | Israel | Randomised control trial | 120 | From 24-28 weeks gestation until birth | Food, insulin and blood glucose values may be uploaded to the app | Monitoring of blood glucose and lifestyle influences |
| 7 | New Zealand | Focus Group | 5 women with GDM 5 Health Professionals | N/A | Variable | Variable |
| 12 | Norway | Interpretative Phenomenological Analysis | 17 | Interviews lasted roughly 30 minutes | Bluetooth blood glucose monitoring | Monitoring of blood glucose. Lifestyle modification information |
Discussion
Overall, the three studies that included health professionals suggested that they were generally satisfied with the use of mobile apps to support the management of GDM. The main issue that was reported by these health professionals was the fact that most of the applications did not have sufficient focus on the particular, person-specific, management that is required in GDM. In particular, they queried that some aspects of management, such as optimal blood glucose levels, require personalised health professional input and some generic information supplied by these apps may not be appropriate in the majority of circumstances. However, they maintained that the line of communication between health professionals and women with GDM that the apps offered could help overcome these issues, particularly if more tailored information could be included in the future.
The feedback from women with GDM was generally positive. These apps allowed for women to take greater initiative with their health and allowed for frequent updates with their respective health professionals. Common themes that were reported included a greater sense of agency and increased confidence in managing their GDM. In some instances, however, a lack of clarity in the information provided by the apps as to what sort of behaviour should be seen as “risky”, such as eating specific food groups or exercise routines, promoted a sense of anxiety in some participants12 . Some also reported that excessive focus on their food intake and blood glucose readings impaired their overall sense of wellbeing 12. These concerns were echoed in a number of the written reviews given for apps identified in the first part of this review, where lack of detailed information was given as a main issue.
Limitations
This review only searched abstract, title, and topics in proposed databases, which may not yield a complete pool of relevant articles. Articles published in other languages (not in English) were also excluded. Furthermore, many of the studies included in this review only involved small numbers of participants, limiting the power of their conclusions. Additionally, some of the apps that were utilised in the studies were not available on the app stores at the time of the search conducted by this review. This was partially due to some of them being web-based applications and thus not available on the stores. Finally, none of the apps reviewed provided support for women speaking languages other than English.
Conclusion
This scoping review describes the perception of women with GDM and health professionals of mobile apps available for managing GDM. We identified and described seven studies that found that overall mobile apps have provided a positive influence in the management of GDM. Future studies should focus on how mHealth technologies can be tailored to women with GDM, how they compare to other forms of information provision and record keeping/sharing, and the impact of their use on maternal and foetal outcomes.
References
1.Nankervis A, Price S, Conn J. Gestational diabetes mellitus. Australian Journal for General Practitioners. 2018;47:445-9.
2.
Moses RG, Morris GJ, Petocz P, San Gil F, Garg D. The impact of potential new diagnostic criteria on the prevalence of gestational diabetes mellitus in Australia. Medical Journal of Australia. 2011;194(7):338-40.
3.
Australia D. Gestational Diabetes in Australia Position Satement. Diabetes Australia; 2020.
4.
Klonoff DC. The Current Status of mHealth for Diabetes: Will it Be the Next Big Thing? Journal of Diabetes Science and Technology. 2013;7(3):749-58.
5.
Arksey H, O’Malley L. Scoping studies: towards a methodological framework. International Journal of Social Research Methodology. 2005;8(1):19-32.
6.
Hong Q, Fâ¡bregues S, Bartlett G, Boardman F, Cargo M, Dagenais P, et al. The Mixed Methods Appraisal Tool (MMAT) version 2018 for information professionals and researchers. Educ Inf. 2018;34:285-91.
7.
Pais S, Parry D, Petrova K, Rowan J. Acceptance of Using an Ecosystem of Mobile Apps for Use in Diabetes Clinic for Self-Management of Gestational Diabetes Mellitus. Stud Health Technol Inform. 2017;245:188-92.
8.
Garnweidner-Holme LM, Borgen I, Garitano I, Noll J, Lukasse M. Designing and Developing a Mobile Smartphone Application for Women with Gestational Diabetes Mellitus Followed-Up at Diabetes Outpatient Clinics in Norway. Healthcare (Basel). 2015;3(2):310-23.
9.
Garnweidner-Holme L, Hoel Andersen T, Sando MW, Noll J, Lukasse M. Health Care Professionals’ Attitudes Toward, and Experiences of Using, a Culture-Sensitive Smartphone App for Women with Gestational Diabetes Mellitus: Qualitative Study. JMIR Mhealth Uhealth. 2018;6(5):e123.
10.
Hirst JE, Mackillop L, Loerup L, Kevat DA, Bartlett K, Gibson O, et al. Acceptability and user satisfaction of a smartphone-based, interactive blood glucose management system in women with gestational diabetes mellitus. J Diabetes Sci Technol. 2015;9(1):111-5.
11.
Miremberg H, Ben-Ari T, Betzer T, Raphaeli H, Gasnier R, Barda G, et al. The impact of a daily smartphone-based feedback system among women with gestational diabetes on compliance, glycemic control, satisfaction, and pregnancy outcome: a randomized controlled trial. Am J Obstet Gynecol. 2018;218(4):453.e1-.e7.
12.
Skar JB, Garnweidner-Holme LM, Lukasse M, Terragni L. Women’s experiences with using a smartphone app (the Pregnant+ app) to manage gestational diabetes mellitus in a randomised controlled trial. Midwifery. 2018;58:102-8.