Hypoglycaemia: Why does it matter?

Introduction
Hypoglycaemia is a common potentially preventable consequence of insulin and insulin secretagogues, such as sulfonylureas. It continues to be a barrier limiting glycaemic management in type 1 (T1D) and type 2 diabetes (T2D).1 Insulin and sulfonylureas are effective in reducing plasma glucose and their actions are independent of glucose levels, which can lead to hypoglycaemia if circulating insulin levels are too high.2 Hypoglycaemia remains a major concern for people living with diabetes, their family and/or support persons and health care professionals.3
Several counter-regulatory stress responses are induced by a hypoglycaemia episode in the effort to restore circulating glucose. Symptoms and physiological effects associated with autonomic stress activation can impact the heart, brain, eyes, and other regulatory responses. Repeated hypoglycaemia episodes can result in impaired counter-regulatory responses, with an increased risk of developing impaired hypoglycaemia awareness.4 This article reviews the mechanisms by which people with insulin-treated diabetes and/or sulfonylureas develop impaired hypoglycaemia awareness as well as the acute and cumulative consequences of hypoglycaemia.
Defining hypoglycaemia
The brain relies on a constant supply of glucose as its primary fuel source to function.5 Hypoglycaemia results from an imbalance between glucose production and glucose utilisation, resultant in glucose falling to less than 4.0 mmol/L5 . For example, hypoglycaemia can occur in instances of elevated circulating insulin (excessive endogenous or exogenous insulin), impaired counter-regulatory regulation, deficiency of counter-regulatory hormones or inability to mobilise and utilise gluconeogenic substrates. Low glucose concentrations are counteracted with a well-orchestrated response to prevent further progression of hypoglycaemia and achieve a prompt recovery in glucose to maintain concentrations between 4.0-8.0 mmol/L.4, 5
Homeostatic responses begin as soon as the glucose reaches approximately 4.0 mmol/L. The evidence for harm associated with impaired cognition, cardiac arrhythmia and mortality increases as glucose levels drop below 3.0 mmol/L.4 For people living with diabetes, hypoglycaemia may be better defined by their clinical symptoms involving the degree of distress and disruption that the episode may cause; this may include the inconvenience of having to consume carbohydrates when not wanting to, and other unpleasant symptoms of the acute response, including confusion, loss of consciousness and possibly, coma.7, 8 This underpins the Consensus Statement from the International Hypoglycaemia Study Group, defining the three levels of plasma glucose when describing hypoglycaemia caused by glucose-lowering therapies (exogenous insulin and/or insulin secretagogues).2, 4 This was endorsed and adopted by the key diabetes organisations; American Diabetes Association (ADA), European Association for the Study of Diabetes (EASD), International Society for Paediatric and Adolescent Diabetes (ISPAD) and the European Medicines Agency (EMA) as well as the international expert panel on continuous glucose monitoring.2, 4, 6 Hypoglycaemia episodes are characterised into three groups as outlined in Table 1.7.
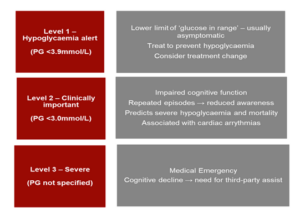
TABLE 1: Classification of hypoglycaemia 7
Landmark randomised control trials such as the Diabetes Control and Complications Trial (DCCT) and the United Kingdom Prospective Diabetes Study (UKPDS), emphasised the importance of tight glycaemic management to prevent the development of microvascular and macrovascular complications.8 Findings from both these trials directed treatment strategies to achieving a target HbA1c ≤ 7% (53 mmol/mol).8 -10 However, it should be noted that the DCCT showed a 3-fold increased risk in severe hypoglycaemia events in participants with T1D in the intervention group compared to usual care. There were more than 1000 episodes of loss of consciousness. However, no deaths, stroke or myocardial infarction were attributed to hypoglycaemia.8, 9 Similarly, the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial showed an increase in all-cause mortality, with a 3-fold increase in risk of hypoglycaemia in high-risk participants with T2D when attempting to achieve aggressive HbA1c (<6.5% / 48mmol/mol) targets.8, 9
Symptoms of hypoglycaemia
Symptoms of hypoglycaemia commonly occur with plasma glucose levels <3 mmol/L. However, the blood glucose levels at which signs and symptoms occur varies among individuals and within an individual at different times.5, 7 For people with diabetes, hypoglycaemia symptoms may vary depending on glycaemic management or previous episodes.5 Symptoms can be divided into neurogenic (autonomic) and neuroglycopenic categories. Neurogenic symptoms result from the sympatho-adrenal release triggered by the hypoglycaemia event. Symptoms such as tremor, palpitations and anxiety are mediated by the activation of the adrenergic system,4, 5, 8 whereas symptoms such as sweating, hunger and tingling are mediated by the activation of the cholinergic system. Neuroglycopenic symptoms result from the brain being deprived of glucose during hypoglycaemia, and may include behavioural changes, fatigue, confusion, visual changes, seizure, and loss of consciousness. The most severe neuroglycopenic symptoms are coma and death.5, 7, 8
Counter-regulatory responses to hypoglycaemia
The counter-regulatory response to hypoglycaemia is a complex process, involving the detection of low glucose, followed by the secretion of several hormones including glucagon, adrenaline, cortisol, and growth hormone to orchestrate recovery of hypoglycaemia.4, 5, 12 In people with intact counter-regulatory responses, a glucose level at 3.6-3.9 mmol/L increases adrenergic and cholinergic neurotransmission in the sympathoadrenal system and the central nervous system.4 The first response is the reduction in pancreatic beta-cell insulin secretion and the release of glucagon. In controlled settings, autonomic symptoms generally develop at the threshold of plasma glucose around 3.2 mmol/L, while neuroglycopenic symptoms and decline in cognitive function have been found to occur at a lower threshold of 2.7 mmol/L.4 Both adrenaline and noradrenaline act on the liver to increase the breakdown of glycogen (glycogenolysis), and lipids and proteins (gluconeogenesis) into glucose when energy production is required. Of note, the glucose levels at which there is activation of catecholamine responses have been shown to be higher in children than in adults and vary depending on glycaemic management.5 The effects of growth hormone and cortisol during hypoglycaemia are delayed compared to the rapid effects of glucagon and adrenaline to regulate glucose.5
Factors affecting normal counter-regulation
There are multiple factors contributing to the intensity of the counter-regulatory response of hypoglycaemia. Glycaemic thresholds can vary significantly depending on the frequency and recency of hypoglycaemia episodes.4 Repeated hypoglycaemia has been shown to blunt the counter-regulatory response.4 A single episode of hypoglycaemia (≤ 3.3mmol/L) has been shown to significantly blunt responses to subsequent episodes of hypoglycaemia.2, 4 Age has also been shown to blunt some facets of the counter-regulatory responses.1, 6, 7, 12 Meneilly et al. showed healthy elderly participants had impaired adrenaline and glucagon responses and reduced awareness of autonomic symptoms compared to healthy younger participants in insulin-induced hypoglycaemia.4 Gender can also influence counter-regulatory responses as evidenced from a study showing female participants with insulin-induced hypoglycaemia had lower secretion of adrenaline, glucagon and growth hormone compared to the male participants, despite all had similar glycaemic thresholds.4
Adrenaline and cortisol responses during early night-time hypoglycaemia can be enhanced.13 There is reduced adrenergic response during sleep, after exercise and in people taking opioids and benzodiazepines.1, 4, 8, 12, 14 Hypoglycaemia in the context of alcohol is a common occurrence where symptoms may not be recognised and hypoglycaemia may occur after several hours, or even the following day.8, 15 In the event of severe hypoglycaemia in those affected by alcohol consumption, the liver continues to break down the alcohol in the system blocking responses to glucagon instead of acting on signals to release more glucagon. In this situation, the administration of glucagon is rendered virtually useless and is not effective in reversing the hypoglycaemia.15
Neuroendocrine responses to hypoglycaemia
Type 1 diabetes
The normal counter-regulatory response to hypoglycaemia is altered in a variety of ways in individuals with T1D. Firstly, due to pancreatic beta-cell failure and the need for exogenous insulin therapy, systemic insulin levels cannot decrease as glucose concentrations fall.4, 6 Secondly, low alpha-cell glucose concentration, which would normally trigger pancreatic alpha-cell glucagon secretion, is compromised due to the loss of interaction between the alpha and beta-cells.4, 5
The increase in adrenaline levels as glucose levels fall is also attenuated, shifting plasma glucose to a lower threshold in people with well-managed T1D.4-6 As a result, adrenergic symptoms fail to appear until the plasma glucose drops to a lower threshold that elicits the adrenaline response. The change in adrenaline response likely results from preceding hypoglycaemia, shifting the thresholds for counter-regulation initiation.4, 5
Type 2 diabetes
There have been limited and conflicting studies on the neuroendocrine responses to hypoglycaemia in people with T2D taking different oral and injectable therapies as well as those who are insulin deficient with low C-peptide levels.4, 11 While many studies have shown that adrenaline responses remained intact, one study found reduced symptomatic and adrenalin responses in participants exposed to insulin-induced hypoglycaemia.4 Variable glucagon responses could be attributed to residual beta-cell function. Although the risk of hypoglycaemia is low in the earlier stages of T2D, hypoglycaemia becomes progressively more frequent in those who develop secondary absolute endogenous insulin deficiency.4 Tight glycaemic management in people with T2D has been shown to shift the threshold for counter-regulatory hormone release to lower glucose concentrations and onset of symptoms of hypoglycaemia, as found in people with T1D.4, 14
Impaired hypoglycaemia awareness
Defective counter-regulation can progress to impaired hypoglycaemia awareness and lead to severe hypoglycaemia resulting in serious harm to the person with diabetes.13 Impaired hypoglycaemia awareness is an acquired disorder associated with insulin treatment and is asserted to result from recurrent exposure to hypoglycaemia episodes (<3 mmol/L).4, 8 In both T1D and T2D, counter-regulatory responses can be perturbed with defective responses potentially leading to impaired hypoglycaemia awareness.12 Glycaemic thresholds for counter-regulatory responses shift to higher glucose concentrations in people with suboptimally managed diabetes and lower glucose concentrations in people who experience recurrent hypoglycaemia.4-6, 8.
Both children and adults with impaired hypoglycaemia awareness have a 6-fold increase of developing severe hypoglycaemia, often missing the opportunity to treat their hypoglycaemia in a timely manner. Further, hypoglycaemia awareness can be affected by sleep, alcohol, and drug use.10 In T2D, the prevalence of impaired hypoglycaemia awareness ranges from 6-17% and is associated with a 9-17-fold increase for severe hypoglycaemia 8, 10, 12 while in T1D the prevalence of impaired hypoglycaemia awareness is 25-40%.8, 10
As impaired hypoglycaemia awareness is potentially caused by recurrent hypoglycaemia, reducing the frequency of hypoglycaemia through appropriate relaxation of glycaemic targets is indicated. Hypoglycaemia awareness may be restored through strict hypoglycaemia avoidance with vigorous monitoring of behavioural modifications.12 These interventions have shown to promote restoration of hypoglycaemia awareness in up to 45% of cases without deterioration in glycaemic management.8, 25 Additionally, continuous glucose monitoring (CGM) and flash glucose monitoring (FGM) systems have been beneficial in reducing the risk, frequency and severity of hypoglycaemia.10, 17 Both CGM and FGM systems can provide a more complete picture of glucose trends and overall glycaemic management, compared to capillary blood glucose monitoring alone.10, 23
The Clarke Hypoglycaemia Awareness Survey and Gold Questionnaire are both validated self-reporting risk scoring instruments to assess for impaired hypoglycaemia awareness. It is important to assess and ask about hypoglycaemia awareness during clinic consultations to identify people with diabetes at high risk.24 It is also crucial to educate family/support persons about what to look out for and how to treat hypoglycaemia in the event of severe hypoglycaemia.6, 8 This should include education about glucagon administration.11, 12
Supportive education about self-monitoring of blood glucose, interpretation of glucose results and education to optimise insulin dosing where appropriate, should be implemented to reduce or eliminate the risk of severe hypoglycaemia and an attempt to regain hypoglycaemia awareness.7, 8
Cumulative impact of hypoglycaemia
Typically, people with diabetes achieve complete recovery following a hypoglycaemia episode, however, there are potential consequences and impacts on overall and future health.3, 8 Psychological impacts include experiencing unpleasant symptoms of stress response, as well as the distress involving interruptions of daily activities, sleep and forced feeding.3 Additionally, a fear of hypoglycaemia may develop and lead to behaviours that negatively impact diabetes management, with a study finding that 40-50% of an adult clinic population with T1D expressed fear of hypoglycaemia.3 Family and support persons are also greatly affected by fear of hypoglycaemia, including parents and carers of children or people living with disability.3, 16 Partners of people with impaired hypoglycaemia awareness and recurrent severe episodes have expressed high levels of distress negatively impacting sleep and quality of life. Societal impacts of hypoglycaemia include restricted employment, loss of driving privileges and relationship breakdown, while economic impacts arise from additional costs associated with healthcare visits and non-direct costs including time lost from employment.8
Physiological effects of hypoglycaemia
The autonomic activation following a hypoglycaemic episode may include an array of symptoms and physiological effects, which can progress from palpitations and sweating, to impaired cognitive function and seizures.3, 8 Depending on the severity and duration of the episode, hypoglycaemia can result in coma and death.8 Hypoglycaemia also can cause short term and long-term effects on the heart, eyes, brain, and inflammatory responses as outlined below.3, 7, 8
Inflammatory Markers
Hypoglycaemia has been shown to be associated with increased inflammatory markers (i.e., C-reactive protein and vascular endothelial growth factor) and a marker of oxidative stress, which increases the risk of endothelial dysfunction in coagulation increasing risk for cardiovascular events lasting several days.3 The ACCORD study identified a link between these proinflammatory changes in people with diabetes, reporting higher rates of adverse cardiovascular events with one or more episodes of hypoglycaemia.9
The eyes
Hypoglycaemia can cause visual disorders such as double vision, dimness or blurred vision and loss of contract sensitivity in people with diabetes. Acute effects can lead to a reduction in central retinal function in humans. Animal studies have also shown retinal degeneration and cone cell death due to induced hypoglycaemia.9
The brain
The brain is reliant on a constant supply of glucose to function.5, 8 Acute interruption of glucose supply can cause reversible and progressive deterioration in cognitive function.8 There is an association between repeated episodes of severe hypoglycaemia and long-term reduction in cognitive function.8 Severe episodes of hypoglycaemia in elderly persons with diabetes has been associated with an increased risk of dementia, functional brain failure and cerebellar ataxia.8, 18
The heart
Acute hypoglycaemia has significant effects on cardiac function and workload. These changes occur via increased heart rate and peripheral systolic blood pressure, fall in central blood pressure and reduction in peripheral arterial resistance, and increased myocardial contractility, stroke volume and cardiac output.8
Adrenaline release combined with excess insulin in circulation during hypoglycaemia, increases uptake of potassium, resulting in low plasma potassium (hypokalaemia). Hypokalaemia is associated with cardiac arrhythmias.3
Electrocardiograph changes can show ectopic activity, flattening of the T-wave, ST depression, ventricular tachycardia, and atrial fibrillation, along with prolonged QT interval and bradycardia.3, 8 QT prolongation is associated with high-risk tachycardia, fibrillation, and sudden cardiac death.8 Sudden death during sleep has been linked with cardiac arrhythmia triggered by nocturnal hypoglycaemia in people with T1D.9
The ACCORD study identified that ventricular tachycardia and fibrillation are promoted by hypoglycaemia, demonstrating that people with diabetes and pre-existing cardiac disease were at an increased risk during an acute hypoglycaemic episode.8
Effects in the elderly person
Elderly people are at risk of harm due to their increased risk of hypoglycaemia.1 Risks may be related to a change in dietary intake and appetite.17 Factors such as deterioration in renal and liver function, which can lead to pharmacokinetic changes with the extension of drug half-life and reduced gluconeogenesis, should be considered. Further, polypharmacy and drug/drug interactions pose an increased risk in this vulnerable population group.20
Glycaemic thresholds triggering counter-regulatory responses to hypoglycaemia are lowered in elderly persons (<2.0 mmol/L), reducing time for corrective action.1 Hypoglycaemia risk is increased with impaired hypoglycaemia awareness and slower reaction time to treat the event.1 The elderly have also been shown to have more prominent neuroglycopenic symptoms, which can result in misdiagnosis such as delirium or other neurological events.21 All these factors need to be carefully considered in the treatment of the older person, with the aim for less ambitious glycaemic targets.1, 12.
In the hospital inpatient setting, elderly people are particularly vulnerable to adverse effects of hypoglycaemia and these are associated with extended length of stay and increased hospital or post discharge mortality.17 Risk factors include continuation of insulin secretagogues, use of sliding scale insulin regimens, and failure to adjust regular insulin doses despite changes in nutritional intake.17
Impact on driving
Driving is a common activity which can be significantly impacted by hypoglycaemia.8, 18 Drivers treated with insulin and/or sulfonylureas are required to demonstrate and understanding of the potential risks of hypoglycaemia and regularly monitor their glucose at times relevant to their driving.8 Drivers should be advised to self-monitor their blood glucose levels before driving, and every two hours on longer trips as practical.19 Hypoglycaemia treatment should also be carried in the vehicle for self-treatment in case of low blood glucose. The safe driving glycaemic target is at or above 5 mmol/L to drive.19
In the event of a severe hypoglycaemic event, the person should be advised not to drive, with a minimum period of six weeks before returning to drive.19 This period of non-driving may depend on many factors such as the reason for the episode, type of vehicle licence and endocrinologist or diabetes specialist opinion. Returning to drive requires a medical clearance and should be based on the drivers behavioural and objective measures to monitor their glucose over a recommended time interval.8, 19 The Austroad Fitness To Drive guidelines were updated in 2016 to recognise the use of CGM or FGM systems or blood glucose monitoring to exclude hypoglycaemia.19 The use of CGM or FGM systems are particularly valuable for those drivers who have impaired hypoglycaemia awareness, with the provision of alerts to warn of impending hypoglycaemia and the opportunity to intervene and prevent the event.
Effect of exercise
Fear of hypoglycaemia remains a significant barrier for people with diabetes to exercise,22 as there is an increased risk of hypoglycaemia due to the increase in glucose utilisation and insulin sensitivity (~3 days) and a blunted counter-regulatory response.8 Hypoglycaemia is more common during endurance and anaerobic exercise (e.g., resistance/weights, sprints and high intensity training), which can attenuate exercise-related lowering of glucose.8 This has been shown during and after exercise in young, healthy adults with T1D.12 Additional carbohydrates may need to be consumed and insulin doses adjusted before and after exercise. It is recommended that glucose levels are monitored closely before, during and after exercise. The use of diabetes technologies such as sensor-augmented or hybrid closed loop insulin pumps, CGM or FGM systems have been shown to be useful in providing alerts to warn of impending hypoglycaemia and/or enable precise adjustment of insulin delivery in these scenarios.10, 23
Experience and perceptions of hypoglycaemia among people with diabetes
It is important to understand that the person with diabetes perception of hypoglycaemia may differ from that of the diabetes care team.11 Experiencing a hypoglycaemic event for the first time can be frightening and may be communicated as being severe.8, 11 The approach of the diabetes care team must include the psychosocial and behavioural aspects of diabetes management and not just focus on glycaemic targets. People with diabetes should be supported through an education program that focuses on problem-solving interventions, to help recognise, assess, and solve problems associated with their diabetes. This problem-solving approach would also guide proactive and preventative self-management strategies to reduce the re-occurrence of hypoglycaemia.11, 12
Treatment of hypoglycaemia
Conscious people with suspected or symptomatic hypoglycaemia should have their blood glucose levels checked. If unable to confirm with blood glucose test, as a precaution they should treat as hypoglycaemia.8 If the person is unconscious, drowsy, and unable to swallow, this is a medical emergency.27


Table 2: Treatment of hypoglycaemia (adults on insulin injections) 8, 27
Prevention of hypoglycaemia
Prevention of hypoglycaemia should be a priority. Empowering people with diabetes and their family/support persons to monitor and be aware of the risks and consequences of hypoglycaemia through effective education and collaboration is vital.8 Risks for hypoglycaemia should be identified and strategies developed to mitigate those risks. Under-reporting and undisclosed reporting of hypoglycaemia may be problematic when assessing for impaired hypoglycaemia awareness.6
Fear of hypoglycaemia and impaired hypoglycaemia awareness should be assessed routinely by health care professionals to identify individuals who need help managing these symptoms. The key focus should be aimed at minimising hypoglycaemia exposure and preventing long-term complications, with hypoglycaemia being a metric for assessing effectiveness of glucose-lowering therapies.4 The provision of an individualised treatment plan and preventative strategies are the key components for reducing risk of hypoglycaemia and safety concerns.8, 11 Strategies may include relaxation of glycaemic targets, de-intensifying, or simplification of glucose-lowering regimens, reducing treatment burden.4, 6, 11
The use of diabetes technologies such as sensor-augmented or hybrid closed loop pump therapy, CGM and FGM systems, allow for real-time notification of glucose values and auditory or vibratory predictive and low glucose alerts. These systems can be used by the person with diabetes independently and have shown to be useful tools for decreasing time spent in hypoglycaemia and prevention of hypoglycaemia.10, 17 Real-time CGM and FGM systems allow for frequent glucose measurement with reduced need for capillary blood glucose monitoring23 and may help detect nocturnal or asymptomatic hypoglycaemia.6 Many diabetes technology companies have cloud-based software programs allowing the person with diabetes to share their glucose data in real-time.6 This allows for remote monitoring of glucose data for the both the person with diabetes and diabetes care team. Remote monitoring enables useful analysis of glucose data through ambulatory glucose profile reports, making it easier to detect glycaemic patterns and variability.6
Conclusion
Hypoglycaemia remains a significant health risk and the greatest barrier to achieving optimal glycaemic management in people with diabetes who are treated with insulin and/or sulfonylureas. The cumulative impact of hypoglycaemia is multifaceted, encompassing many short- and long-term health implications. Exposure to recurrent hypoglycaemia is an important factor in the pathogenesis of impaired hypoglycaemia awareness. Larger studies are needed to explore and understand the benefits of new diabetes technologies, as well as behavioural and educational interventions focusing on the prevention of hypoglycaemia and improvement of hypoglycaemia awareness to reduce fear of hypoglycaemia.
Key points
- Hypoglycaemia prevention must remain the key focus in supporting diabetes management and treatment to reduce harm to the person with diabetes.
- People with diabetes and their family/support persons should be counselled on situations that may increase risk of hypoglycaemia e.g., delayed meals, during and after alcohol consumption, during and after exercise.
- Fear and impaired hypoglycaemia awareness should be assessed on a routine basis to identify individuals who require support and intervention.
- Collaboration between the person living with diabetes and their family/support persons is vital to develop and adjust individualised treatment plans.
- Sensor-augmented or hybrid closed loop pump therapy, CGM and FGM systems are useful tools for decreasing time spent in hypoglycaemia and prevention of hypoglycaemia.
Acknowledgements
I would like to thank Associate Professor Elif Ekinci, Professor Andrea Driscoll and Anna Peters for their expert advice and suggestions.
References
1.Morales J, Schneider D. Hypoglycemia. Am J Med. 2014 Oct;127(10 Suppl): S17-24. doi:
2.de Galan BE, McCrimmon RJ, Ibberson M, et al. Reducing the burden of hypoglycaemia in people with diabetes through increased understanding: design of the Hypoglycaemia REdefining SOLutions for better liVEs (Hypo-RESOLVE) project. Diabet Med. 2020;37(6):1066-1073. doi:10.1111/dme.14240
3.Amiel SA. The consequences of hypoglycaemia. Diabetologia. 2021 May;64(5):963-970. doi: 10.1007/s00125-020-05366-3. Epub 2021 Feb 7. PMID: 33550443; PMCID: PMC8012317.
4.Tesfaye N, Seaquist ER. Neuroendocrine responses to hypoglycemia. Ann N Y Acad Sci. 2010 Nov; 1212:12-28. doi: 10.1111/j.1749-6632.2010.05820. x. Epub 2010 Oct 29. PMID: 21039590; PMCID: PMC2991551.
5.Sprague JE, Arbeláez AM. Glucose counterregulatory responses to hypoglycemia. Pediatr Endocrinol Rev. 2011 Sep;9(1):463-73; quiz 474-5. PMID: 22783644; PMCID: PMC3755377.
6.Kenny C. When hypoglycemia is not obvious: diagnosing and treating under-recognized and undisclosed hypoglycemia. Prim Care Diabetes. 2014 Apr;8(1):3-11. doi: 10.1016/j.pcd.2013.09.002. Epub 2013 Oct 5. PMID: 24100231.
7.American Diabetes Association. 6. Glycemic Targets: Standards of Medical Care in Diabetes-2020. Diabetes Care. 2020 Jan;43(Suppl 1): S66-S76. doi: 10.2337/dc20-S006. PMID: 31862749.
8.Kalra S, Mukherjee JJ, Venkataraman S, Bantwal G, Shaikh S, Saboo B, Das AK, Ramachandran A. Hypoglycemia: The neglected complication. Indian J Endocrinol Metab. 2013 Sep;17(5):819-34. doi: 10.4103/2230-8210.117219. PMID: 24083163; PMCID: PMC3784865.
9.Frier BM, Schernthaner G, Heller SR. Hypoglycemia and cardiovascular risks. Diabetes Care. 2011 May;34 Suppl 2(Suppl 2): S132-7. doi: 10.2337/dc11-s220. PMID: 21525444; PMCID: PMC3632150.
10.Lin YK, Fisher SJ, Pop-Busui R. Hypoglycemia unawareness and autonomic dysfunction in diabetes: Lessons learned and roles of diabetes technologies. J Diabetes Investig. 2020 Nov;11(6):1388-1402. doi: 10.1111/jdi.13290. Epub 2020 Jul 7. PMID: 32403204; PMCID: PMC7610104.
11.Silbert R, Salcido-Montenegro A, Rodriguez-Gutierrez R, Katabi A, McCoy RG. Hypoglycemia Among Patients with Type 2 Diabetes: Epidemiology, Risk Factors, and Prevention Strategies. Curr Diab Rep. 2018 Jun 21;18(8):53. doi: 10.1007/s11892-018-1018-0. PMID: 29931579; PMCID: PMC6117835.
12.MartÃn-Timón I, Del Cañizo-Gómez FJ. Mechanisms of hypoglycemia unawareness and implications in diabetic patients. World J Diabetes. 2015 Jul 10;6(7):912-26. doi: 10.4239/wjd. v6. i7.912. PMID: 26185599; PMCID: PMC4499525.
13.Diabetes Canada Clinical Practice Guidelines Expert Committee, Yale JF, Paty B, Senior PA. Hypoglycemia. Can J Diabetes. 2018 Apr;42 Suppl 1: S104-S108. doi: 10.1016/j.jcjd.2017.10.010. PMID: 29650081.
14.MartÃn-Timón I, Del Cañizo-Gómez FJ. Mechanisms of hypoglycemia unawareness and implications in diabetic patients. World J Diabetes. 2015 Jul 10;6(7):912-26. doi: 10.4239/wjd. v6. i7.912. PMID: 26185599; PMCID: PMC4499525.
15.Beyond Type 1. 2021. Why doesn’t glucagon work with alcohol? [online] Available at: https://beyondtype1.org/why-doesnt-glucagon-work-with-alcohol. Accessed 10 November 2021
16.Haugstvedt A, Wentzel-Larsen T, Aarflot M, Rokne B, Graue M. Assessing fear of hypoglycemia in a population-based study among parents of children with type 1 diabetes – psychometric properties of the hypoglycemia fear survey – parent version. BMC Endocr Disord. 2015; 15:2. Published 2015 Jan 19. doi:10.1186/1472-6823-15-2
17.Gosmanov AR, Mendez CE, Umpierrez GE. Challenges and Strategies for Inpatient Diabetes Management in Older Adults. Diabetes Spectr. 2020;33(3):227-235. doi:10.2337/ds20-0008
18.Bree AJ, Puente EC, Daphna-Iken D, Fisher SJ. Diabetes increases brain damage caused by severe hypoglycemia. Am J Physiol Endocrinol Metab. 2009;297(1): E194-E201. doi:10.1152/ajpendo.91041.2008
19.Austroads.com.au. 2021. Assessing Fitness to Drive â 3.2.1 Hypoglycaemia. [online] Available at: https://austroads.com.au/publications/assessing-fitness-to-drive/ap-g56/diabetes-mellitus/general-assessment-and-management7r5-mgn/hypoglycaemia [accessed 5 December 2021]
20.Freeman J. Management of hypoglycemia in older adults with type 2 diabetes. Postgrad Med. 2019;131(4):241-250. doi:10.1080/00325481.2019.1578590
21.Chow L, Seaquist ER. How Significant Is Severe Hypoglycemia in Older Adults With Diabetes? Diabetes Care. 2020;43(3):512-514. doi:10.2337/dci19-0069
22.Cigrovski Berkovic M, Bilic-Curcic I, La Grasta Sabolic L, Mrzljak A, Cigrovski V. Fear of hypoglycemia, a game changer during physical activity in type 1 diabetes mellitus patients. World J Diabetes. 2021;12(5):569-577. doi:10.4239/wjd. v12.i5.569
23.Triki N, Yekutiel N, Levi L, Azuri J. The effects of continuous glucose monitoring system on patient outcomes and associated costs in a real-world setting. Diabet Med. 2021;38(5): e14518. doi:10.1111/dme.14518
24.Ghandi K, Pieri B, Dornhorst A, Hussain S. A Comparison of Validated Methods Used to Assess Impaired Awareness of Hypoglycaemia in Type 1 Diabetes: An Observational Study. Diabetes Ther. 2021;12(1):441-451. doi:10.1007/s13300-020-00965-0
25.Yeoh E, Choudhary P, Nwokolo M, Ayis S, Amiel SA. Interventions That Restore Awareness of Hypoglycemia in Adults With Type 1 Diabetes: A Systematic Review and Meta-analysis. Diabetes Care. 2015;38(8):1592-1609. doi:10.2337/dc15-0102
26.Andreozzi F, Candido R, Corrao S, et al. Clinical inertia is the enemy of therapeutic success in the management of diabetes and its complications: a narrative literature review. Diabetol Metab Syndr. 2020; 12:52. Published 2020 Jun 17. doi:10.1186/s13098-020-00559-7
27.Ndss.com.au. 2021. Managing hypoglycaemia fact sheet â NDSS. [online] Available at: https://www.ndss.com.au/about-diabetes/resources/find-a-resource/managing-hypoglycaemia-fact-sheet. Accessed 10 November 2021