Bariatric Surgery for the Management of Type 2 Diabetes: A Review

Introduction
Obesity is a major risk factor for the development of T2 Diabetes (T2D) with a 3-fold increased prevalence of diabetes in obese individuals and over 90% of people with diabetes are obese.1, 2
Almost two-thirds (63.4%) of Australian adults were overweight or obese in 2014-15.3 The prevalence of obesity and T2D are rising in a mirrored fashion, leading to the term ‘diabesity’.4 Various hypotheses including inflammation, adipokines and lipid overflow have been proposed to explain the causal links between obesity and insulin resistance.4 A simplified relationship is shown in Figure 1.5 Bariatric surgery is the single most effective treatment for severe obesity with significantly better and sustained weight loss compared to lifestyle or pharmacological therapies.6
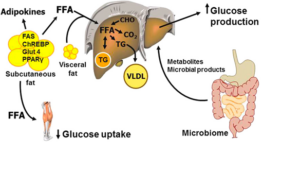 Figure 1: The pathophysiology of obesity. (Cefalu WT et al, 2015).5
Figure 1: The pathophysiology of obesity. (Cefalu WT et al, 2015).5
Bariatric surgery as a therapeutic option for diabetes
Bariatric surgery has emerged as a highly effective treatment for T2D, especially in its capacity to induce ‘diabetes remission’,1, 6-8 defined by the American Diabetes Association as normal fasting blood glucose levels (<7.0mmol/L) or normal HbA1c (<6.5%) without pharmacologic therapy for at least one year.1 Almost all individuals with T2D will benefit from improved glycaemic management occurring post bariatric surgery, and reduction in diabetes medication requirements often precede the weight loss, with many people no longer requiring insulin post-surgery.9
Diabetes remission rates vary widely across studies, dependent upon the bariatric surgical procedures, duration of diabetes (better outcomes for those who have been diagnosed less than 4 years), c-peptide levels, age and insulin dependence (better outcomes for those managed with diet or oral therapy only).1 Bariatric surgery can also be used for primary prevention of diabetes. In the Swedish Obesity Study (SOS) trial, surgery decreased the risk of developing T2D by 96% at 2 years and by 78% at 15 years.1
Eligibility criteria
Historically, a body mass index (BMI) ≥40kg/m2 or a BMI≥35 kg/m2 with an obesity related comorbidity (such as diabetes) were the accepted indication for bariatric surgery. However, evidence from more recent studies such as the STAMPEDE trial, show that people with lower BMIs (down to BMI 27kg/m2) also show benefit.1, 10 A consensus statement from the 2nd Diabetes Surgery Summit in 2016 recommends bariatric surgery in those with a BMI 30.0-34.9kg/m2 and suboptimal glycaemic management despite optimal medical management of diabetes.1, 7 The Queensland State-wide Bariatric Service (QSBS) uses a current HbA1c >6.5% despite treatment with at least two medications and a BMI greater than 35kg/m2 (maximum weight 185 kg) as inclusion criteria.11
Traditionally, the exclusion criteria for bariatric surgery included severe end-organ damage such as cirrhosis with portal hypertension, severe heart disease, lung disease, and chronic kidney disease (CKD) with eGFR < 30ml/min.7, 9 QSBS and many others also exclude those with active smoking, drug or alcohol dependency, malignancy and unstable mental health conditions.7, 9, 11
However with careful selection bariatric surgery continues to benefit overall outcomes in early cirrhosis, heart failure and even advanced CKD, although the magnitude of benefit (% excess weight loss) can be reduced in those with CKD stage IV or V.12-14 Age alone is also not a contraindication as older individuals seem to have comparable weight loss and T2D remission rates despite a higher risk of complications.15
The psychosocial health of surgical candidates also need to be considered. Obesity itself is associated with a relatively higher prevalence of psychopathological conditions and poorer quality of life.16, 17 Health related quality of life and mental health generally improve after bariatric surgery, but some people experience negative psychological reactions post-surgery, and most bariatric surgical programs require psychological assessment as part of their selection criteria, as pre-surgical psychosocial factors appear important for postoperative mental health.18
Selection of bariatric surgery technique
Most bariatric surgical techniques can now be done laparoscopically and are classified into restrictive and malabsorptive (Fig 2) according to their effect on the gastrointestinal anatomy and function.9, 19 Restrictive-type procedures physically reduce gastric volume and caloric intake inducing early satiety via postulated neurohormonal regulation. Vertical sleeve gastrectomy (VSG) is the prototype of this group wherein 70-80% of the stomach is resected, resulting in a tubular remnant with significantly reduced capacity and enhanced gastric emptying. Due to the relative simplicity, minimal complications and good clinical outcomes, VSG has increased exponentially over the last two decades making it the most common bariatric surgery worldwide. It accounted for 61% of bariatric surgeries in 2018 with a T2D remission rate of up to 65%.19 Adjustable gastric banding (AGB) involves placing a silicone ring in the upper stomach. This procedure has been rapidly phasing out (less than 10% of surgeries in 2018) since its benefit on weight loss is relatively limited, with increased complications risk and poorer T2D remission rates (33% at 1 year).19
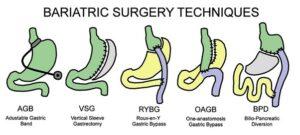
Figure 2: Various techniques for bariatric surgery.
Malabsorptive surgeries work by promoting macronutrient malabsorption. Biliopancreatic diversion (BPD) establishes a bypass of the small bowel to induce bile acid and food to be mixed in the distal 50-100cm of the ileum. BPD is considered to be the most effective of all the bariatric surgeries for weight loss and diabetes remission (95% remission rate at 2 years), but due to its technical complexity and higher complication rates (especially nutrient deficiencies), the use of BPD surgery has been declining, and only accounted for 1% of total bariatric surgeries in 2018.19
Mixed restrictive and malabsorptive procedures like Roux-en-Y gastric bypass (RYGB) and the more recent one-anastomosis gastric bypass (OAGB) combine gastric reduction and malabsorption induced by a bowel bypass. They have better weight loss efficacy, but also slightly increased risk of complications including nutrient deficiencies. T2D remission rates after RYGB have been reported to be 60% at 1 year and 75% at 2 years. RYGB surgeries accounted for 17% of bariatric procedures in 2018.15 OAGB involves a single site anastomosis between the remnant gastric pouch and a jejunal omega loop, also termed a mini-gastric bypass. This surgery is simpler with less morbidity with initial studies showing equivalent or better weight loss and metabolic outcomes than those associated with RYGB. However, there may be an increased risk of biliary reflux, anastomotic ulcers and more nutritional deficiencies necessitating more long-term data before widespread acceptance.20, 21
Preoperative medical management for bariatric surgery
Many individuals with obesity have pre-existing vitamin and mineral deficiencies. Due to the increased risk of micronutrient deficiency after surgery it is important to screen for and correct these deficiencies as well as optimise glycaemic managementpre-operatively.6, 8 Most bariatric surgery protocols include a low-calorie diet leading unto surgery, since a modest weight loss of 5-10% immediately prior seems to lower perioperative complications.6, 22 In the enhanced recovery after surgery (ERAS) protocol, prior weight loss is encouraged to help early functional recovery and early discharge. Stefura et al, showed a preoperative weight loss of ≥5% was associated with improved short-term weight loss after surgery, but did not otherwise influence the perioperative course.23 Presently, evidence for preoperative weight loss influencing postoperative long-term weight loss outcomes is controversial. However, this should not be a sole contraindication to proceeding with surgery.24
Complications & sequelae of bariatric surgery
Bariatric surgery has a low mortality rate (0.04-0.3%), with a 4.3% incidence of a major adverse events in the early postoperative period indicating significant improvements in safety over the past 20 years.9 Surgical complications include sepsis (often from anastomotic dehiscence), shock (due to postoperative haemorrhage), or cardiopulmonary events, with thromboembolic disease being the leading cause of death (incidence of 0.34%).9 Later surgical complications are often procedure-specific including anastomotic stricture formation, ulceration and incisional and internal herniation.9
Hypoglycaemia
With rapid improvement in glycaemic management following surgery, early postoperative hypoglycaemia can often be due to continuation of glucose lowering medications (particularly sulfonylureas or insulin), requiring prompt discontinuation or significant dose reduction of these agents.25
Early dumping syndrome, mainly associated with RYGB procedures, is caused by the rapid emptying of nutrients into the small bowel, raising osmolality, which triggers a cascade of gut hormonal changes. This leads to vasomotor and gastrointestinal symptoms, characterised by nausea, tremor, sweats, diarrhoea, flushing, tachycardia, dizziness and sometimes syncope. These symptoms occur immediately after eating (within 15 minutes of a meal) and affect up to 50% of people who have undergone a mixed or malabsorptive procedure usually within the first 3 months after surgery.8, 9, 23 Treatment relies predominantly upon diet modification (avoidance of rapid carbohydrates and alcohol) with small and frequent meals (around 6 per day) and delaying fluid intake until 30 minutes after a meal. Stomach emptying may be further delayed by lying down for 30 min after eating.22
Postprandial hyperinsulinaemic hypoglycaemia (also termed late dumping syndrome) is far less common, manifesting between 3-36 months after surgery, with 10% of people developing significant hypoglycaemia 1-3 hours after meals.26 It is triggered by altered nutrient delivery and pancreatic / enteral hormonal axes.8 Symptoms of hypoglycaemia such as tremor, perspiration, fatigue, weakness, confusion, hunger and syncope with low blood glucose levels and response to glucose (Whipple’s triad) are required for diagnosis. A mixed meal tolerance test is the most appropriate diagnostic test.8 In the absence of a predictive tool to identify those at risk, prior counselling is integral to treatment.8, 22 Dietary modification remains the mainstay of treatment including replacement of high glycaemic Index (GI) foods by balanced diets (low GI high fibre carbohydrates, lean protein and reduced saturated fat). Alcohol and excessive caffeine should be avoided as they impair hepatic glucose release. More than two-thirds of individuals with late dumping syndrome will achieve symptomatic relief by dietary changes8 whilst the rest will require adjunctive pharmacological treatment with acarbose, metformin or rarely somatostatin analogues.8, 22 GLP1-analogues are also used anecdotally. However, the use of pharmacological therapies is often mitigated by side effects and limited efficacy.8, 22
Bone health
There is increased bone turnover and bone loss following all bariatric surgeries, but an increased risk of fractures has only been consistently reported after malabsorptive-type surgeries (such as BPD). Several factors including decreased mechanical loading, secondary hyperparathyroidism from vitamin D and/or calcium deficiency etc have been implicated. Individuals should be counselled on the benefits of physical activity after surgery, especially for their bone health.8, 27
Nutritional consequences
All bariatric surgeries can potentially cause or aggravate significant micronutrient deficiencies and all surgical candidates require nutritional assessment and management pre-and postoperatively by an Accredited Practising Dietitian with experience in bariatric surgery.6, 8 Postoperative nutritional monitoring is lifelong, usually falling to primary care physicians beyond the initial two years.6, 8, 22 Deficiencies in vitamin D, iron and vitamin B12 are common to all bariatric procedures whilst malabsorptive surgeries can also cause deficiencies in calcium, zinc, copper, selenium and other fat-soluble vitamins. All individuals who have undergone bariatric surgery should commence on multivitamin and mineral supplements, choice and dosing being determined by any pre-existing deficiencies and the extent of malabsorption expected from the surgery.6, 8 Malabsorptive bariatric surgery increases the risk of nephrolithiasis due to fat malabsorption leading to calcium oxalate stones with a prevalence of 22-28%. Generous hydration is the mainstay of therapy, but often challenging, due to the restricted gastric volumes.8
Other complications
Other complications include gallstone formation and hair thinning.9 Non-specific abdominal pain can occur in 5-10% of individuals, the diagnosis and treatment of which can be challenging.9
Symptoms of nausea, bloating and early satiety can also cause people to struggle to tolerate their often significantly increased ‘pill burden’ post-surgery (including micronutrient supplementation as well as diabetes medications).28
Alcohol metabolism is altered after bariatric surgery, resulting in earlier and higher blood alcohol concentrations, and it is important that people are counselled regarding this.9 There is also a subset of individuals who have an increased risk of alcohol misuse after surgery, particularly after RYGB. Predictive variables include being male, younger age, smoking, regular alcohol consumption, preoperative alcohol use disorder, and a lower sense of belonging.29
Although most people experience improvements in their mental health and quality of life following bariatric surgery, a minority of people experience a deterioration, often driven by unrealistic expectations preoperatively and pre-existing psychopathology.17 Some suffer from ongoing body image dissatisfaction, of which excess skin as a consequence of the weight loss may also contribute.17 Weight regain is also associated with increased depression.17 Eating behaviour disorders can occur postoperatively, including rigid eating control due to a continuous fear of regaining weight.17 This is more common in those with preoperative eating disorders, which is seen in 5-15% of bariatric surgery candidates.17
Pregnancy after bariatric surgery
A significant number of women undergoing bariatric surgery are of child-bearing age.30 Fertility rates may improve post bariatric surgeries, although these procedures are not currently endorsed as a treatment for infertility in obese women.8, 27 Guidelines suggest delaying pregnancy for 12-18 months to ensure weight stabilisation and adequate nutrition prior to conception.6, 8 Vitamin A supplements during pregnancy should be in the form of beta-carotene rather than retinol palmitate due to concerns of teratogenicity in the first trimester. All women should have a full nutritional screen preconception and then every trimester.6, 8 VSG and RYGB are both associated with lower rates of gestational diabetes mellitus (GDM), non-elective caesarean sections, and large for gestational age (LGA) infants, but with increased risk for small for gestational age infants and need for iron supplementation. RYGB is also associated with mildly higher rates of preterm birth.8
Screening for GDM in these women is a challenge as the oral glucose tolerance test (OGTT ) is poorly tolerated. As per Shaw et al 2019, current practice uses capillary blood glucose profile for 1 week between 24 -28 weeks of gestation as a practical alternative with the same targets as for the general population26 . At present, there are no specific guidelines for gestational weight gain or breast feeding in this cohort.26
Long-term outcomes of bariatric surgery
Weight loss from bariatric surgery is significant and durable, with malabsorptive procedures performing better than restrictive procedures.9 A meta-analysis of published long-term outcomes (10 or more years post-surgery) showed the most significant weight loss is seen in BPD (+/- duodenal switch) with 71% excess weight loss (EWL), followed by RYGB with 60% EWL, and finally AGB with 49% EWL. Data was insufficient for a meta-analysis of sleeve gastrectomy outcomes, but two smaller studies reveal a weighted mean of 57% EWL.31 Few studies have followed bariatric surgery outcomes beyond 20+ years. The CBS longitudinal cohort study showed maximal weight loss by two years postoperatively and a relatively constant effect over the next 18 years, with a final EWL of 48.9% and total weight loss (TWL) of 22.2% at 20 years.31 As for diabetes, more than 50% of those in remission will relapse; however, the years of remission are known to have a remarkable legacy effect over the subsequent decades in terms of micro-/macrovascular disease burden.1 Data from the SOS trial, reported a drop in diabetes remission rates from 72.4% at 2 years to 38.1% at 10 years and 30.4% at 15 years after surgery. The main predictors of relapse were longer duration of diabetes and insulin use prior to the surgery.1 An overwhelming 95% of people who have undergone bariatric surgery have reported improved quality of life with a 30-40% reduced mortality at 10 years.9
Revisional bariatric surgery
Revisional bariatric surgery is becoming more common, and 5-8% of primary bariatric operations will require a revision procedure.32 The leading reasons for revision include inadequate weight loss or specific inherent complications related to the primary surgical procedure. Conversion of a restrictive-type procedure to a RYGB is the gold standard option. There is an increase in morbidity and mortality rates for revisional surgeries compared to primary operations, but the additional weight loss achieved and correction of any initial surgical complications often justify this risk.9, 32
Conclusion
- ‘Diabesity’ is increasing in prevalence in younger age groups and bariatric surgery is a clinically viable and cost-effective treatment both in terms of quality-adjusted life-year (QALY) and mortality benefits.
- The outcomes of bariatric surgery are highly dependent on appropriate candidate selection, type of procedure, prior duration and severity of diabetes and good long-term care.
- In general VSG remains the most common procedure worldwide due to its minimal complications risk with very reasonable weight loss and diabetes remission benefits.
- Malabsorptive procedures are gradually becoming safer and more acceptable with significantly improved long-term diabetes remission and weight loss efficacy, although closer follow up is often needed for micronutrient deficiencies.
- Counselling and allied health support is crucial to avoid confusion and disengagement, particularly in regards to expectations of benefit, risks, side effects such as postoperative hypoglycaemia, avoidance of alcohol and preconception care.
References
1.Affinati AH, Esfandiari NH, Oral EA, Kraftson AT. Bariatric Surgery in the Treatment of Type 2 Diabetes. Curr Diab Rep. 2019;19(12):156.
2.
Institute BIHD. Diabetes: the silent pandemic and its impact on Australia. https://www.diabetesaustralia.com.au/wp-content/uploads/Diabetes-the-silent-pandemic-and-its-impact-on-Australia.pdf. Published 2012. Accessed 7th November, 2021.
3.Huse O, Hettiarachchi J, Gearon E, Nichols M, Allender S, Peeters A. Obesity in Australia. Obes Res Clin Pract. 2018;12(1):29-39.
4.
Alexandra Chadt SS, Hans-Georg Joost, Hadi Al-Hasani. Molecular links between Obesity and Diabetes: âDiabesityâ. 2018. https://www.ncbi.nlm.nih.gov/books/NBK279051/. Published Updated 2018 Jan 23.
5.Cefalu WT, Bray GA, Home PD, et al. Advances in the Science, Treatment, and Prevention of the Disease of Obesity: Reflections From a Diabetes Care Editors’ Expert Forum. Diabetes Care. 2015;38(8):1567-1582.
6.
O’Kane M, Parretti HM, Pinkney J, et al. British Obesity and Metabolic Surgery Society Guidelines on perioperative and postoperative biochemical monitoring and micronutrient replacement for patients undergoing bariatric surgery-2020 update. Obes Rev. 2020;21(11):e13087.
7.Arterburn DE, Telem DA, Kushner RF, Courcoulas AP. Benefits and Risks of Bariatric Surgery in Adults: A Review. Jama. 2020;324(9):879-887.
8.
Collazo-Clavell ML, Shah M. Common and Rare Complications of Bariatric Surgery. Endocrinol Metab Clin North Am. 2020;49(2):329-346.
9.
le Roux CW, Heneghan HM. Bariatric Surgery for Obesity. Med Clin North Am. 2018;102(1):165-182.
10.
Schauer PR, Bhatt DL, Kirwan JP, et al. Bariatric Surgery versus Intensive Medical Therapy for Diabetes – 5-Year Outcomes. N Engl J Med. 2017;376(7):641-651.
11.Standard QH-DoH. Statewide Bariatric Service. Queensland Government. https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwiZ477wtpn0AhU4ILcAHSdNBtcQFnoECAQQAQ&url=https%3A%2F%2Fwww.health.qld.gov.au%2F__data%2Fassets%2Fpdf_file%2F0019%2F1012591%2Fqh-imp-481.pdf&usg=AOvVaw374VdI-6gVs5yYyIu2WAwx. Published 2020. Updated 10 November 2020. Accessed.
12.Rodriguez Flores M, Aguilar Salinas C, Piché ME, Auclair A, Poirier P. Effect of bariatric surgery on heart failure. Expert Rev Cardiovasc Ther. 2017;15(8):567-579.
13.
Hansel B, Arapis K, Kadouch D, et al. Severe Chronic Kidney Disease Is Associated with a Lower Efficiency of Bariatric Surgery. Obes Surg. 2019;29(5):1514-1520.
14.
Jan A, Narwaria M, Mahawar KK. A Systematic Review of Bariatric Surgery in Patients with Liver Cirrhosis. Obes Surg. 2015;25(8):1518-1526.
15.
Haywood C, Sumithran P. Treatment of obesity in older persons-A systematic review. Obes Rev. 2019;20(4):588-598.
16.Jumbe S, Hamlet C, Meyrick J. Psychological Aspects of Bariatric Surgery as a Treatment for Obesity. Curr Obes Rep. 2017;6(1):71-78.
17.
Kubik JF, Gill RS, Laffin M, Karmali S. The impact of bariatric surgery on psychological health. J Obes. 2013;2013:837989.
18.
Wimmelmann CL, Dela F, Mortensen EL. Psychological predictors of mental health and health-related quality of life after bariatric surgery: a review of the recent research. Obes Res Clin Pract. 2014;8(4):e314-324.
19.
Jin ZL, Liu W. Progress in treatment of type 2 diabetes by bariatric surgery. World J Diabetes. 2021;12(8):1187-1199.
20.Robert M, Espalieu P, Pelascini E, et al. Efficacy and safety of one anastomosis gastric bypass versus Roux-en-Y gastric bypass for obesity (YOMEGA): a multicentre, randomised, open-label, non-inferiority trial. Lancet. 2019;393(10178):1299-1309.
21.
Parikh M, Eisenberg D, Johnson J, El-Chaar M. American Society for Metabolic and Bariatric Surgery review of the literature on one-anastomosis gastric bypass. Surg Obes Relat Dis. 2018;14(8):1088-1092.
22.
Bettini S, Belligoli A, Fabris R, Busetto L. Diet approach before and after bariatric surgery. Rev Endocr Metab Disord. 2020;21(3):297-306.
23.Stefura T, DroÅ J, Kacprzyk A, et al. Influence of Preoperative Weight Loss on Outcomes of Bariatric Surgery for Patients Under the Enhanced Recovery After Surgery Protocol. Obes Surg. 2019;29(4):1134-1141.
24.
Krimpuri RD, Yokley JM, Seeholzer EL, Horwath EL, Thomas CL, Bardaro SJ. Qualifying for bariatric surgery: is preoperative weight loss a reliable predictor of postoperative weight loss? Surg Obes Relat Dis. 2018;14(1):60-64.
25.
Vouri SM, Chen J, Sparkman J, Salles A, Micek ST. Order of discontinuation of glucose-lowering medications following bariatric surgery. Diabetes Res Clin Pract. 2021;172:108580.
26.
Shawe J, Ceulemans D, Akhter Z, et al. Pregnancy after bariatric surgery: Consensus recommendations for periconception, antenatal and postnatal care. Obes Rev. 2019;20(11):1507-1522.
27.Casimiro I, Sam S, Brady MJ. Endocrine implications of bariatric surgery: a review on the intersection between incretins, bone, and sex hormones. Physiol Rep. 2019;7(10):e14111.
28.
Kennedy AL, Nelson T, Pettine S, Miller BF, Hamilton KL, Donovan EL. Medication use following bariatric surgery: factors associated with early discontinuation. Obes Surg. 2014;24(5):696-704.
29.
Ivezaj V, Benoit SC, Davis J, et al. Changes in Alcohol Use after Metabolic and Bariatric Surgery: Predictors and Mechanisms. Curr Psychiatry Rep. 2019;21(9):85.
30.
Falcone V, Stopp T, Feichtinger M, et al. Pregnancy after bariatric surgery: a narrative literature review and discussion of impact on pregnancy management and outcome. BMC Pregnancy Childbirth. 2018;18(1):507.
31.
O’Brien PE, Hindle A, Brennan L, et al. Long-Term Outcomes After Bariatric Surgery: a Systematic Review and Meta-analysis of Weight Loss at 10 or More Years for All Bariatric Procedures and a Single-Centre Review of 20-Year Outcomes After Adjustable Gastric Banding. Obes Surg. 2019;29(1):3-14.
32.
Switzer NJ, Karmali S, Gill RS, Sherman V. Revisional Bariatric Surgery. Surg Clin North Am. 2016;96(4):827-842.