The Enteral Enigma: managing diabetes and enteral nutrition
Introduction
Diabetes is now the fastest growing chronic condition in Australia.1 Individuals with a diagnosis of diabetes are known to have a threefold higher risk of hospitalisation.2 Many of the sequelae of suboptimally managed diabetes such as stroke can give rise to the need for enteral feeding. Furthermore, enteral nutrition combined with the stress response seen in acute illness can increase the risk of hyperglycaemia in those who were previously normoglycemic.3
Factors contributing to stress-induced hyperglycaemia which impair glucose uptake in the peripheral tissues include:
- elevated production of counter regulatory hormones
- reduced production of insulin and increased hepatic glucose production, secondary to insulin resistance
- proteolysis (protein breakdown) and adipose tissue lipolysis (fat breakdown)
The result of this is a range of immune changes, haemodynamic and tissue effects as shown in Figure 1, translating into increased risk of infection and impaired wound healing which can ultimately lead to prolonged length of stay and death.
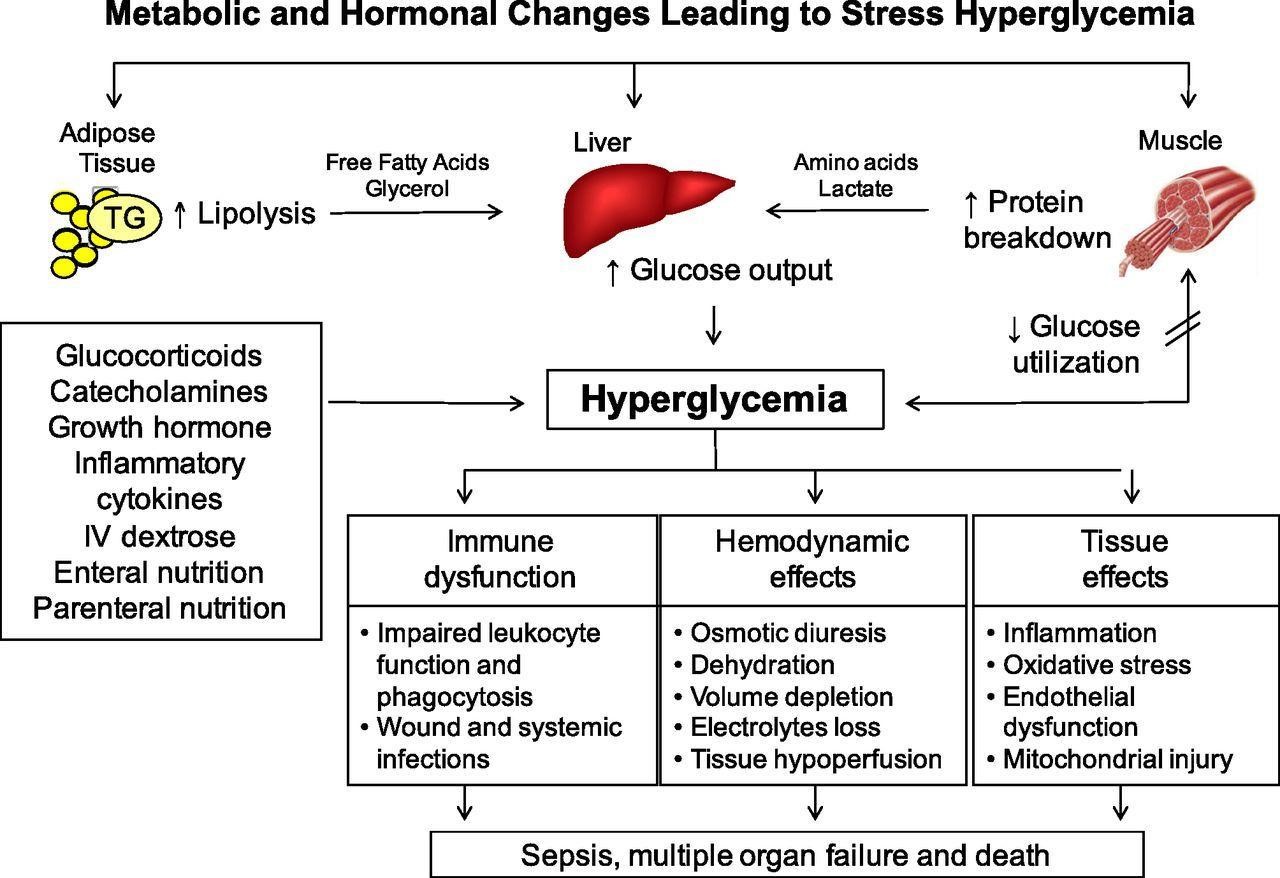
Figure 1: Mechanisms leading to stress-induced hyperglycaemia41
Given the direct relationship between the acute phase response and impaired glycemic management, it is unsurprising to find that hyperglycemia is common within the hospital population. Observational studies worldwide, have reported a prevalence rate of hyperglycemia in those with pre-existing diabetes ranging from 38-40% within the general hospital population and 70-80% within the ICU setting or for those undergoing cardiac surgery.4-7 Specific to Australia, point prevalence surveys have shown 20-25% of people with a pre-existing diagnosis of diabetes develop hyperglycemia during a hospital admission and of these, 40-50% will require insulin.7-9 Stress-induced hyperglycemia in those without pre-existing diabetes typically resolves once the acute illness or stress subsides. However, it has been estimated that at one year post the acute episode, up to 60% of people will have confirmed diabetes, indicating this is far from a benign occurrence.10
Does short term hyperglycemia matter?
There is a substantial body of evidence from observational and prospective randomised controlled trials in people both with and without diabetes that indicates a strong association between hyperglycemia and adverse clinical outcomes including infectious complications, increased length of hospital stay and mortality. 4, 11-15 While most of this evidence was initially derived from research within the critical care population, an increasing number of trials have identified links between hyperglycaemia and increased morbidity and mortality within the broader hospital setting, including people admitted with community acquired pneumonia, those undergoing general or cardiac surgery and individuals admitted to community hospitals.4, 11, 16-18 Of interest, a growing number of studies suggest there is an increased risk of complications in those with stress-induced hyperglycemia compared to those with pre-existing diabetes – indicating optimisation of blood glucose levels during the acute illness period should be prioritised by health practitioners for this vulnerable population.4, 14, 19
Glycemic targets – what do we aim for?
Recommendations differ depending on the population and setting, specifically critical care compared to non-critical care. It should also be noted, that the evidence is relatively weak and weaker still for those receiving enteral feeds. Individualisation is important when deciding on glycemic targets. For example, comorbidities, previous hypoglycaemic events and the level of nursing care available should all be considered when setting targets.
Critical Care recommendations
| Organisation | Guidance |
| American Diabetes Association (ADA)/ American Association of Clinical Endocrinologist (AACE)20 | ● Initiate insulin therapy for persistent hyperglycemia (blood glucose >10 mmol/L) ● Treatment goal: For most people, target blood glucose levels between 7.8-10mmol/L ● More stringent goals (6.1-7.8mmol/L) may be appropriate for selected individuals, if achievable without significant risk of hypoglycaemia |
| American College of Physicians (ACP)21 | ● Recommends against intensive insulin therapy in those with or without diabetes in surgical/medical critical care units ● Treatment goal: target blood glucose between 7.8-11.1mmol/L, in those with or without diabetes, in surgical/medical critical care units |
| Critical care society22 | ● Blood glucose levels of >8.3 mmol/L should trigger insulin therapy ● Treatment goal: maintain blood glucose <8.3mmol/L for most adults in critical care ● Maintain blood glucose levels <10 mmol/L while avoiding hypoglycaemia |
| Society of thoracic surgeons – specific to cardiac surgery23 | ● Continuous insulin infusion preferred over subcutaneous or intermittent intravenous boluses ● Treatment goal: Recommend blood glucose <10 mmol/L during surgery (≤6.1mmol/L in fasting and pre-prandial states) |
General hospital population
| Organisation | General hospital patients |
| ADA/AACE20 | ● No specific guidelines ● If treated with insulin, pre-prandial blood glucose targets should generally be <7.8mmol/L, with random blood glucose levels <10mmol/L ● More stringent targets may be appropriate for those with previously tight glycemic management ● Less stringent targets may be appropriate in those with severe comorbidities |
| Endocrine society24 | ● Pre-prandial blood glucose target <7.8mmol/L and random blood glucose <10 mmol/L ● A lower target range may be appropriate in those able to achieve and maintain glycemic management without hypoglycaemia ● Blood glucose <10 -11.1mmol/L is appropriate in those with terminal illness and/or limited life expectancy, or those at high risk of hypoglycaemia ● Adjust glycemic therapy when glucose falls <5.5mmol/L to avoid hypoglycaemia |
| Joint British Diabetes Society25 | ● Target blood glucose levels in most people between 6- 10 mmol/L, with an acceptable range of between 4-12mmol/L |
| Diabetes UK Position Statement- Enteral feeding of people with stroke and diabetes26 | ● Fasting/pre-prandial 5–8 mmol/L. ● Feeding 6–12 mmol/L
|
| ASPEN27 | ● During feeding: 7.8-10mmol /L |
Selecting the right enteral feed
For decades nutritional companies have worked on developing a range of diabetes-specific formulas (DSF). These formulas contain increased amounts of fibre, reduced percentages of carbohydrate and additional monounsaturated fatty acids (MUFAs), with the aim of reducing the glycemic response in people receiving enteral nutrition. The efficacy of DSF to improve glycemic management has been replicated in several studies and a meta-analysis, however the overall quality of the research remains poor.28-30 In particular the studies have been found to have a high risk of bias, small numbers, short study duration and lack robust data to show improvements in morbidity and mortality.30 As a result, at present both ASPEN and Diabetes UK recommend against using DSF in those receiving EN as an effective means to manage hyperglycemia.25, 27 On the other hand, due to the perceived consistency in improving glycemic outcomes the ESPEN expert group endorses using DSF in individuals considered high risk , namely those with a history of diabetes or obesity.31
A pragmatic approach seems to be to adopt a step-wise process. Start off using a standard polymeric formula and if hyperglycemia persists, adjust insulin/medication regimens. Also consider additional medications that may be impacting on glycemic management such as steroids and review energy targets to ensure avoidance of overfeeding. Once all of these options have been explored, then a DSF could be considered. Due to the increased costs of DSF compared to standard polymeric feeds, if no improvement in glycemic response is found then the person should be switched back to the standard feed. It is also important to note that many of the DSF are lower in protein and should not be considered as a first line choice, especially in critical care situations, where protein requirements are increased. It is widely accepted that meeting nutritional targets should not be compromised for the sake of glycemic management, especially given the acute illness response.
Along with the choice of enteral feeds, the method of delivery is of importance, as this will influence the insulin regimen and the required frequency of blood glucose monitoring. Enteral feeds can be delivered continuously, intermittently (for less than 24 hours) or bolus (where a syringe is used to infuse the formula over a period of usually 15-30 minutes several times per day, often mimicking something closer to a normal eating pattern). The method of delivery will depend on the individual, taking into account factors such as positioning and aspiration risk, clinical requirements for breaks to engage in therapy or rehab, individual preferences, gastric tolerance and others.
Insulin regimens
The evidence for various insulin regimens is limited and so far comes only from trials looking at continuous enteral feeding. Therefore, recommendations for bolus and nocturnal/intermittent regimens are based on expert consensus. In general, intravenous insulin infusion offers the best option for glycemic management. However, this requires intensive input on the part of healthcare teams and in the ward setting is often not practical. The table below summarises recommendations based on existing trials and expert consensus from the literature.3, 32-35
| Setting | Enteral feeding type | Insulin |
| Critical care | All | Intravenous insulin As the person transitions to being ward ready they will typically transition to one of the regimens outlined below using a combination of basal/bolus insulin. |
| Non-critical care | Continuous
| Premixed (70/30) human insulin at the time of feed commencement and every 8 hours with the addition of short acting correction scale bolus insulin every 8 hours. |
| Isophane insulin given every 8 hours (40-50% of total daily dose ), in addition to short/rapid acting insulin (50-60%) plus rapid acting- short acting correction scale every 4-6 hours. | ||
| Continue pre-existing basal bolus insulin. Give the basal insulin at the start of the feed and the bolus dose of soluble or rapid acting analogue at 6 and 12 hours into the feed as required. This is particularly relevant for those with type 1 diabetes (T1D). | ||
| Intermittent or nocturnal tube feeds | Long acting or biphasic 70/30 insulin administered at commencement of feed. Correction or short acting administered as required to achieve glycemic targets. | |
| Bolus | Single dose of human insulin 20 minutes prior to bolus. * For those with T1D basal insulin must be continued. It should also be continued for those with type 2 diabetes already on basal insulin pre-admission. |
Additional practice points:
- For individuals with T1D it is important to continue basal insulin even if the enteral feed is discontinued.
- Generally avoid administering insulin towards the end of the feeding period as this increases the risk of hypoglycaemia.
- For those who are fed during the day there is an increased risk of nocturnal hypoglycaemia especially if intermediate or long acting insulin is used and this should be considered in selecting the type of insulin and deciding on the timing of insulin doses.
- Always take into account the individuals’ oral intake – this may need to be covered by additional insulin.
Monitoring guidelines3, 26
| Type of enteral feeding regimen | Frequency of monitoring |
| Continuous | 4-6 hourly when receiving subcutaneous insulin and continuous feed |
| Intermittent | Pre-feed, 4-6 hourly during feed and 2 hour post-feed |
| Bolus | Pre-feed, 2 hour post-feed and 4-6 hourly if prolonged breaks between feeds |
| Practice Points: ● Beware of hypoglycaemia during fasted period between feeds. In the event of hypoglycaemia the frequency of monitoring should be increased to assess intervention and response. ● If the feed is stopped suddenly and insulin has been given it is essential to be aware of the risk of hypoglycaemia and monitor appropriately. If long acting insulin has been given, consider commencing intravenous 10% dextrose to prevent hypoglycaemia and provide a supply of carbohydrate until enteral feeding can be re-established. If using a variable rate insulin infusion, which is common in critical care, then blood glucose should be monitored hourly.
| |
Hypoglycaemia management
Several studies have shown that hypoglycaemia is associated with adverse outcomes in hospitalised individuals, including increased length of stay, increased risk of death at one year and an association with adverse cardiovascular outcomes.36-39 Similar to hyperglycemia, the exact mechanisms underlying the relationship between hypoglycaemia and adverse outcomes remain unclear. As per ESPEN and the ADA, hypoglycaemia is defined as a blood glucose level of less than 3.9mmol/L.31, 40 There are a wide range of factors that can contribute to hypoglycaemia in individuals receiving enteral feeding including incorrect insulin doses, interruption to feeds for procedures and displacement of feeding tubes. Timely recognition of hypoglycaemia via regular blood glucose monitoring is especially important in those on enteral feeds as many have decreased levels of consciousness and are unable to alert staff to symptoms of hypoglycaemia.
Management of hypoglycaemia in individuals receiving enteral nutrition is complicated by the fact that many are Nil By Mouth (NBM) and therefore treatment must be suitable to be given via the enteral access device or intravenously.
See the Figure 2 outlining a sample treatment algorithm for those NBM on enteral feeds:
 Figure 2: Treatment algorithm for management of hypoglycemia
Figure 2: Treatment algorithm for management of hypoglycemia
Conclusion
Key take home points:
- Hyperglycemia is common in hospitalised individuals due to the acute stress response and is associated with increased risk of morbidity and mortality.
- Glycemic management and insulin requirements will change throughout an admission as the stress response changes.
- Diabetes-specific formulas should not be considered as first line management for hyperglycemia. Standard enteral formulas are usually appropriate for those with pre-existing diabetes and stress-induced hyperglycemia.
- Never compromise nutritional adequacy for glycemic management. Once overfeeding has been ruled out then review medical management (e.g. has the insulin regimen been optimised?; are any non-essential medications affecting blood glucose levels?)
- When deciding on appropriate insulin regimen and frequency of blood glucose monitoring, consider the type and timing of enteral feeding regimen (continuous, intermittent or bolus).
- Prevent hypoglycaemia by having in place appropriate protocols if enteral feeds are ceased for procedures or tubes are dislodged.
- Specific protocols should be in place for both critical care and the general ward setting, to guide management of individuals on enteral feeds and insulin. These protocols should address glucose monitoring, insulin types and adjustments, hypoglycaemia management and prompting referrals to endocrine/diabetes services.
- Given the interplay of enteral feed choices and glycemic management, good communication between dietitians and endocrine/diabetes teams is paramount to drive proactive rather than reactive blood glucose management and to optimise outcomes.
Translation to practice
78 year old male with insulin requiring, admitted with acute respiratory failure due to influenza
Admitted to the intensive care unit (ICU) and intubated.
Usual home insulin regimen Novomix 30 bd (60u mane and 70u nocte), with HbA1c 7.1%
Hyperglycemia in ICU managed by insulin infusion – eventually weaned to Lantus 80u bi-daily (BD) prior to ward.
Transferred to ward with enteral nutrition (EN) running via nasogastric tube (NGT), and tolerating small amounts of texture modified diet.
NGT dislodged over weekend, and deemed not for reinsertion – for oral diet trial as per the medical team.
Lantus dose not reviewed and prescribed 80u nocte dose received, resulting in hypoglycaemia to 2.4mmol/L overnight
Endocrine team review the following morning and regimen reduced to 20u Lantus BD whilst oral intake slowly improving.
Lessons learned:
Insulin requirements reduce as critical illness resolve – need for regular review and adjustment at time of stepdown to ward level and when carbohydrate intake changes in timing or amount, especially if sudden and unplanned.
Importance of having specific protocols for management of blood glucose levels and when there are unplanned interruptions of nutritional delivery.
Acknowledgements
For kindly reviewing prior to publication: Madeleine Neff APD.
References
1.Australia D. Diabetes in Australia Canberra, Australia Diabetes Australia 2015 [Available from: https://www.diabetesaustralia.com.au/diabetes-in-Australia.
2.Welfare AIoHa. Hospital care for diabetes Canberra, Australia Australian Institute of Health and Welfare; 2019 [Available from: https://www.aihw.gov.au/reports/diabetes/diabetes-snapshot/contents/hospital-care-for-diabetes.
3.Drincic AT, Knezevich JT, Akkireddy P. Nutrition and Hyperglycemia Management in the Inpatient Setting (Meals on Demand, Parenteral, or Enteral Nutrition). Curr Diab Rep. 2017;17(8):59.
4.Umpierrez GE, Isaacs SD, Bazargan N, You X, Thaler LM, Kitabchi AE. Hyperglycemia: an independent marker of in-hospital mortality in patients with undiagnosed diabetes. J Clin Endocrinol Metab. 2002;87(3):978-82.
5.Carpenter DL, Gregg SR, Xu K, Buchman TG, Coopersmith CM. Prevalence and Impact of Unknown Diabetes in the ICU. Crit Care Med. 2015;43(12):e541-50.
6.Schmeltz LR, DeSantis AJ, Thiyagarajan V, Schmidt K, O’Shea-Mahler E, Johnson D, et al. Reduction of surgical mortality and morbidity in diabetic patients undergoing cardiac surgery with a combined intravenous and subcutaneous insulin glucose management strategy. Diabetes Care. 2007;30(4):823-8.
7.Bach L EE, Engler D, Gilfillan C, Hamblin S, MacIsaac RJ, Soldatos G, Steele C, Ward GM, Wyatt S. The high burden of inpatient diabetes mellitus: the Melbourne Public Hospitals Diabetes Inpatient Audit. Med J Aust. 2014;201:334-8.
8.Lan NSR, Li C, Fegan PG. Diabetes prevalence is high in hospital patients: a Western Australia perspective. Internal Medicine Journal. 2019;49(4):551-2.
9.Taylor JE, Campbell LV, Zhang L, Greenfield JR. High diabetes prevalence and insulin medication errors in hospital patients. Internal Medicine Journal. 2018;48(12):1529-32.
10.Van Ackerbroeck S, Schepens T, Janssens K, Jorens PG, Verbrugghe W, Collet S, et al. Incidence and predisposing factors for the development of disturbed glucose metabolism and DIabetes mellitus AFter Intensive Care admission: the DIAFIC study. Crit Care. 2015;19:355.
11.Baker EH, Janaway CH, Philips BJ, Brennan AL, Baines DL, Wood DM, et al. Hyperglycaemia is associated with poor outcomes in patients admitted to hospital with acute exacerbations of chronic obstructive pulmonary disease. Thorax. 2006;61(4):284-9.
12.Capes SE, Hunt D, Malmberg K, Pathak P, Gerstein HC. Stress hyperglycemia and prognosis of stroke in nondiabetic and diabetic patients: a systematic overview. Stroke. 2001;32(10):2426-32.
13.Bruno A, Gregori D, Caropreso A, Lazzarato F, Petrinco M, Pagano E. Normal glucose values are associated with a lower risk of mortality in hospitalized patients. Diabetes care. 2008;31(11):2209-10.
14.Falciglia M, Freyberg RW, Almenoff PL, D’Alessio DA, Render ML. Hyperglycemia-related mortality in critically ill patients varies with admission diagnosis. Crit Care Med. 2009;37(12):3001-9.
15.Krinsley JS. Association between hyperglycemia and increased hospital mortality in a heterogeneous population of critically ill patients. Mayo Clin Proc. 2003;78(12):1471-8.
16.Ramos M, Khalpey Z, Lipsitz S, Steinberg J, Panizales MT, Zinner M, et al. Relationship of perioperative hyperglycemia and postoperative infections in patients who undergo general and vascular surgery. Ann Surg. 2008;248(4):585-91.
17.Noordzij PG, Boersma E, Schreiner F, Kertai MD, Feringa HH, Dunkelgrun M, et al. Increased preoperative glucose levels are associated with perioperative mortality in patients undergoing noncardiac, nonvascular surgery. Eur J Endocrinol. 2007;156(1):137-42.
18.Furnary AP, Gao G, Grunkemeier GL, Wu Y, Zerr KJ, Bookin SO, et al. Continuous insulin infusion reduces mortality in patients with diabetes undergoing coronary artery bypass grafting. J Thorac Cardiovasc Surg. 2003;125(5):1007-21.
19.Kotagal M, Symons RG, Hirsch IB, Umpierrez GE, Dellinger EP, Farrokhi ET, et al. Perioperative hyperglycemia and risk of adverse events among patients with and without diabetes. Ann Surg. 2015;261(1):97-103.
20.Moghissi ES, Korytkowski MT, DiNardo M, Einhorn D, Hellman R, Hirsch IB, et al. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes care. 2009;32(6):1119-31.
21.Qaseem A, Humphrey LL, Chou R, Snow V, Shekelle P. Use of intensive insulin therapy for the management of glycemic control in hospitalized patients: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2011;154(4):260-7.
22.Jacobi J, Bircher N, Krinsley J, Agus M, Braithwaite SS, Deutschman C, et al. Guidelines for the use of an insulin infusion for the management of hyperglycemia in critically ill patients. Crit Care Med. 2012;40(12):3251-76.
23.Lazar HL, McDonnell M, Chipkin SR, Furnary AP, Engelman RM, Sadhu AR, et al. The Society of Thoracic Surgeons practice guideline series: Blood glucose management during adult cardiac surgery. Ann Thorac Surg. 2009;87(2):663-9.
24.Umpierrez GE, Hellman R, Korytkowski MT, Kosiborod M, Maynard GA, Montori VM, et al. Management of hyperglycemia in hospitalized patients in non-critical care setting: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2012;97(1):16-38.
25.Group JBDSIC. Joint British Diabetes Societies Inpatient Care Group. Guidelines for the management of inpatient diabetes. 2014 [Available from: https://abcd.care/joint-british-diabetes-societies-jbds-inpatient-care-group.
26.Roberts AW, Penfold S. Glycaemic management during the inpatient enteral feeding of people with stroke and diabetes. Diabet Med. 2018;35(8):1027-36.
27.McMahon MM, Nystrom E, Braunschweig C, Miles J, Compher C. A.S.P.E.N. clinical guidelines: nutrition support of adult patients with hyperglycemia. JPEN J Parenter Enteral Nutr. 2013;37(1):23-36.
28.Elia M, Ceriello A, Laube H, Sinclair AJ, Engfer M, Stratton RJ. Enteral nutritional support and use of diabetes-specific formulas for patients with diabetes: a systematic review and meta-analysis. Diabetes Care. 2005;28(9):2267-79.
29.Ojo O, Brooke J. Evaluation of the role of enteral nutrition in managing patients with diabetes: a systematic review. Nutrients. 2014;6(11):5142-52.
30.Laesser CI, Cumming P, Reber E, Stanga Z, Muka T, Bally L. Management of Glucose Control in Noncritically Ill, Hospitalized Patients Receiving Parenteral and/or Enteral Nutrition: A Systematic Review. J Clin Med. 2019;8(7):935.
31.Barazzoni R, Deutz NEP, Biolo G, Bischoff S, Boirie Y, Cederholm T, et al. Carbohydrates and insulin resistance in clinical nutrition: Recommendations from the ESPEN expert group. Clin Nutr. 2017;36(2):355-63.
32.Korytkowski MT, Salata RJ, Koerbel GL, Selzer F, Karslioglu E, Idriss AM, et al. Insulin therapy and glycemic control in hospitalized patients with diabetes during enteral nutrition therapy: a randomized controlled clinical trial. Diabetes care. 2009;32(4):594-6.
33.Cook A, Burkitt D, McDonald L, Sublett L. Evaluation of glycemic control using NPH insulin sliding scale versus insulin aspart sliding scale in continuously tube-fed patients. NCP – Nutrition in Clinical Practice. 2009;24(6):718-22.
34.Hsia E, Seggelke SA, Gibbs J, Rasouli N, Draznin B. Comparison of 70/30 biphasic insulin with glargine/lispro regimen in non-critically ill diabetic patients on continuous enteral nutrition therapy. Nutr Clin Pract. 2011;26(6):714-7.
35.Vennard K, Selen D, Gilbert M. The management of hyperglycaemia in noncritically ill hospitalised patients treated with continuous enteral or parenteral nutrition NUTRITION. Endocrine Practice. 2018;24(10):900-6.
36.Turchin A, Matheny ME, Shubina M, Scanlon JV, Greenwood B, Pendergrass ML. Hypoglycemia and clinical outcomes in patients with diabetes hospitalized in the general ward. Diabetes Care. 2009;32(7):1153-7.
37.Krinsley JS, Grover A. Severe hypoglycemia in critically ill patients: risk factors and outcomes. Crit Care Med. 2007;35(10):2262-7.
38.Investigators TN-SS. Hypoglycemia and Risk of Death in Critically Ill Patients. New England Journal of Medicine. 2012;367(12):1108-18.
39.Gill GV, Woodward A, Casson IF, Weston PJ. Cardiac arrhythmia and nocturnal hypoglycaemia in type 1 diabetes–the ‘dead in bed’ syndrome revisited. Diabetologia. 2009;52(1):42-5.
40.American Diabetes A. 15. Diabetes Care in the Hospital: Standards of Medical Care in Diabetes-2019. Diabetes Care. 2019;42(Suppl 1):S173-s81.
41.Umpierrez GE, Pasquel FJ. Management of Inpatient Hyperglycemia and Diabetes in Older Adults. Diabetes Care. 2017;40(4):509-17.