Heart Failure in People with Type 2 Diabetes
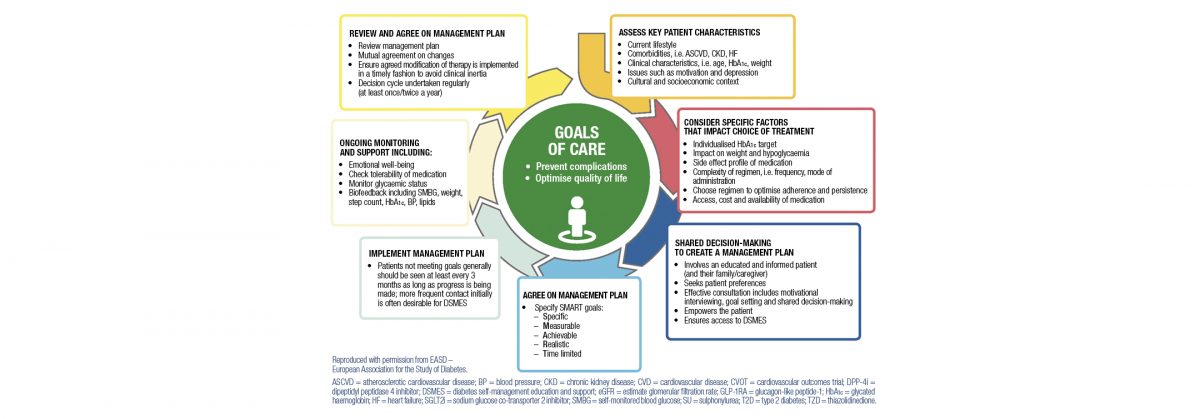
Traditionally, we have adopted a glucocentric approach to the management of diabetes and focused primarily on glycaemic management. Large studies have shown that optimal glucose management reduces the risk of microvascular complications (retinopathy and nephropathy), but the benefits on macrovascular events (myocardial infarction, stroke) have been less pronounced,1 although long-term follow-up studies have shown a small decrease in major cardiovascular (CV) events.2 This is disappointing, as most of the morbidity and mortality associated with type 2 diabetes (T2D) is due to CV events.3, 4 It has become clear that optimal management of blood pressure and lipids, particularly with angiotensin-converting-enzyme inhibitors (ACEI) or angiotensin II receptor blockers (ARB) and statins, has reduced the risk of CV events and renal dysfunction in people with T2D,5–7. Cardiorenal protection is now seen to be as important.8 Until recently, the agents we have used to manage hyperglycaemia in diabetes have not had any impact on reducing CV or renal events, so the advent of the sodium-dependent glucose cotransporters inhibitors (SGLT2i) and glucagon-like peptide-1 agonists (GPL1) have been met with great enthusiasm.
It is often not appreciated that there is an increased risk of heart failure (HF) in people with T2D.9 In fact, people with T2D have a 2-3-fold increased risk of developing HF,10 and there is an increased risk of CV death in these individuals.11–13 T2D is also associated with an increased risk of hospitalisation for HF, whether the person with T2D has HF with reduced ejection fraction (HFrEF) or HF with preserved ejection fraction (HFpEF).12–15 The DPP4 inhibitors are neutral with respect to cardiorenal protection, but there has been a signal of increased hospitalisation for HF with several agents.16 The GLP1 agonists have been shown to confer cardiorenal protection in several large outcome studies, but may have no effect on HF.17–20
In Australia, the threshold for renal function for PBS reimbursement is estimated glomerular filtration rate (eGFR) >45mL/min/1.73m² for Empaglifozin and Dapagliflozin.21, 22 The renal threshold with Ertugliflozin is >45mL/min/1.73m². 23 HbA1C needs to be >7.0% for these agents to be PBS eligible.21, 22, 24, 25
It is essential to educate people about good genital hygiene when considering SGLT2i treatment to reduce the risk of genital mycotic infection,8 and to instruct them to discuss transient treatment interruption in the setting of systemic illness or surgery/procedures where fasting is required (e.g. angiography, colonoscopy, etc) to avoid euglycemic diabetic ketoacidosis (DKA).8 A reduction in diuretic dose also needs to be considered if individuals are on thiazide or loop diuretics (as the SGLT2i’s have a weak diuretic effect).8 Hospitalisation for HF is common in T2D and is associated with considerable morbidity and mortality, in addition to expense to the health care system and socially.8, 12–15 The appropriate use of SGLT2i’s may reduce this risk in people with T2D with established CV disease and should be increasingly be used in these individuals.8
Acknowledgements
This article is commissioned by AstraZeneca.
PLEASE CLICK HERE TO REVIEW FULL PRODUCT INFORMATION BEFORE PRESCRIBING. FURTHER INFORMATION AVAILABLE FROM ASTRAZENECA ON 1800 805 342 OR www.astrazeneca.com.au/PI.
FORXIGA®, XIGDUO®, and QTERN® are registered trademarks of the AstraZeneca group of companies. Registered user AstraZeneca Pty. Ltd. ABN 54 009 682 311. 66 Talavera Road, Macquarie Park, NSW 2113. www.astrazeneca.com.au. For Medical Information enquiries: 1800 805 342 or medinfo.australia@astrazeneca.com. To report an adverse event: 1800 805 342 or via https://aereporting.astrazeneca.com.
AU-6639. 15734. September 2019.
References
1.Huang D et al. BioMed Res Inter 2017; 2017.
2.Holman RR et al. N Engl J Med 2008; 359(15):1577-1589.
3.Seshasai SR et al. N Engl J Med 2011; 364:829-841.
4.International Diabetes Federation (IDF).
IDF Diabetes Atlas, 7th edition.
Brussels, Belgium: International Diabetes Federation, 2015.
5.Liu Y et al. BMC Nephrology 2017; 18:206.
6.Kobori H et al. Curr Pharm Des 2013; 19(17):3033-3042.
7.Strauss MH et al. Can J Diabetes 2018; 42:124-9.
8.American Diabetes Association & European Association for the Study of Diabetes.
Management of hyperglycaemia in type 2 diabetes, 2018.
A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD).
Available at: https://link.springer.com/content/pdf/10.1007%2Fs00125-018-4729-5.pdf Last accessed: September 2019.
9.Lehrke M et al. Am J Cardio 2017; 120(1):S37-47.
10.Kenny HC et al. Circulation Res 2019; 124(1):121-141.
11.Einarson TR et al. Cardiovasc Diabetol 2018; 17:83.
12.Dunlay SM et al. Circulation 2019; 140(7):e294-324.
13.Ofstad AP et al. Heart Failure Revs 2018; 23(3):303-323.
14.Rosengren A et al. Diabetologia 2018; 61(11):2300-9.
15.Targher G et al. Euro J Heart Fail 2017; 19(1):54-65.
16.Scirica et al. N Engl J Med 2013; 369:1317-1326.
17.Marso SP et al. N Engl J Med 2016; 375:311-322.
18.Marso SP et al. N Engl J Med 2016; 375:1834-1844.
19.Scheen AJ. Diabetes Metabolism 2017; 43:2S13-9.
20.Holman RR et al. New Eng J Med 2017; 377(13):1228-1239.
21.Forxiga Approved Product Information.
22.Jardiance Approved Product Information.
23.Stelgatro Approved Product Information.
24.Pharmaceutical Benefits Scheme (PBS). Dapagliflozin. Available at: http://www.pbs.gov.au/medicine/item/10011X Last accessed: September 2019.
25.Pharmaceutical Benefits Scheme (PBS). Empagliflozin. Available at: https://www.pbs.gov.au/medicine/item/10202Y-10206E-11281R-11314L Last accessed: September 2019.
